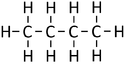Difference between revisions of "Butane"
(→About Butane) |
|||
| Line 29: | Line 29: | ||
: [[Butane]] can be [[oxidise]]d to [[product|produce]] [[Carbon Dioxide]] and [[Water]]. | : [[Butane]] can be [[oxidise]]d to [[product|produce]] [[Carbon Dioxide]] and [[Water]]. | ||
: [[Butane]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | : [[Butane]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | ||
| − | : < | + | : 2C<sub>4</sub>H<sub>10</sub> + 13O<sub>2</sub> → 8CO<sub>2</sub> + 10H<sub>2</sub>O |
Revision as of 12:51, 7 June 2019
Key Stage 3
Meaning
Butane is a gaseous (at room temperature) hydrocarbon with chemical formula CH4.
About Butane
- Butane is hydrocarbon because it contains only Hydrogen and Carbon atoms.
- Butane can be oxidised to produce Carbon Dioxide and Water.
- Butane + Oxygen → Carbon Dioxide + Water
Key Stage 4
Meaning
Butane is a gaseous (at room temperature) alkane with chemical formula CH4.
About Butane
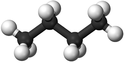
| Chemical Formula (CnH2n+2) | Structural Formula | Structural Diagram | Ball and Stick Model |
| C4H10 | CH3CH2CH2CH3 |
- Butane is hydrocarbon because it contains only Hydrogen and Carbon atoms.
- Butane can be oxidised to produce Carbon Dioxide and Water.
- Butane + Oxygen → Carbon Dioxide + Water
- 2C4H10 + 13O2 → 8CO2 + 10H2O