Difference between revisions of "Deuterium"
(Created page with "==Key Stage 4== ===Meaning=== right|300px|thumb|The [[Chemical Symbol|chemical symbol for Deuterium.]] Deuterium is an isotope of Hy...") |
(No difference)
|
Revision as of 11:50, 31 March 2019
Key Stage 4
Meaning
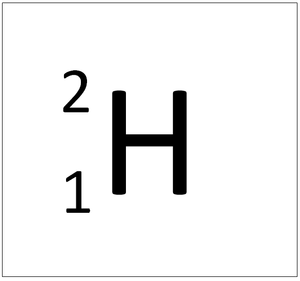
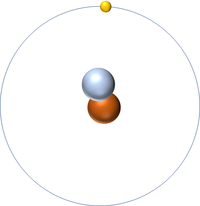
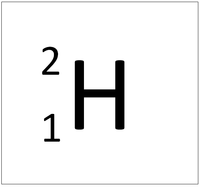
Deuterium is an isotope of Hydrogen containing one neutron.
About Deuterium
- Deuterium has all the same chemical properties of Hydrogen.
- Deuterium is a stable isotope.
| Hydrogen always has 1 proton but in this isotope there is 1 neutron. This isotope of Hydrogen is known as Deuterium. | Hydrogen always has 1 proton but in this isotope there is 2 neutrons. This isotope of Hydrogen is known as Tritium. | |