Difference between revisions of "Deuterium"
| Line 12: | Line 12: | ||
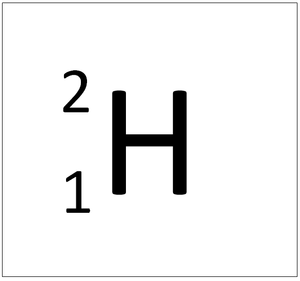
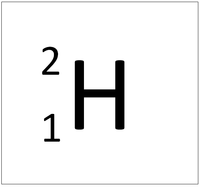
| style="height:20px; width:200px; text-align:center;" colspan = "2"|[[Hydrogen]] always has 1 [[proton]] but in this [[isotope]] there is 1 [[neutron]]. This [[isotope]] of [[Hydrogen]] is known as [[Deuterium]]. | | style="height:20px; width:200px; text-align:center;" colspan = "2"|[[Hydrogen]] always has 1 [[proton]] but in this [[isotope]] there is 1 [[neutron]]. This [[isotope]] of [[Hydrogen]] is known as [[Deuterium]]. | ||
|} | |} | ||
| + | |||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/0008158770/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158770&linkCode=as2&tag=nrjc-21&linkId=ec31595e720e1529e49876c3866fff6e ''Deuterium, page 109, GCSE Physics; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Deuterium, pages 27, 100, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
Latest revision as of 00:02, 4 November 2019
Key Stage 4
Meaning
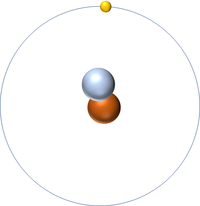
Deuterium is an isotope of Hydrogen containing one neutron.
About Deuterium
- Deuterium has all the same chemical properties of Hydrogen.
- Deuterium is a stable isotope.
| Hydrogen always has 1 proton but in this isotope there is 1 neutron. This isotope of Hydrogen is known as Deuterium. | |