Difference between revisions of "Methane"
| (5 intermediate revisions by 2 users not shown) | |||
| Line 7: | Line 7: | ||
: [[Methane]] can be [[oxidise]]d to [[product|produce]] [[Carbon Dioxide]] and [[Water]]. | : [[Methane]] can be [[oxidise]]d to [[product|produce]] [[Carbon Dioxide]] and [[Water]]. | ||
: [[Methane]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | : [[Methane]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | ||
| − | |||
: [[Methane]] is a [[Greenhouse Gas]] contributing to [[Global Warming]]. | : [[Methane]] is a [[Greenhouse Gas]] contributing to [[Global Warming]]. | ||
| − | ==Key Stage | + | ==Key Stage 4== |
===Meaning=== | ===Meaning=== | ||
| − | [[Methane]] is a [[gas]]eous (at [[Room Temperature|room temperature]]) [[ | + | [[Methane]] is a [[gas]]eous (at [[Room Temperature|room temperature]]) [[alkane]] with [[Chemical Formula|chemical formula]] CH<sub>4</sub>. |
===About Methane=== | ===About Methane=== | ||
| Line 31: | Line 30: | ||
: [[Methane]] can be [[oxidise]]d to [[product|produce]] [[Carbon Dioxide]] and [[Water]]. | : [[Methane]] can be [[oxidise]]d to [[product|produce]] [[Carbon Dioxide]] and [[Water]]. | ||
: [[Methane]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | : [[Methane]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | ||
| − | : < | + | : CH<sub>4</sub> + 2O<sub>2</sub> → CO<sub>2</sub> + 2H<sub>2</sub>O |
: [[Methane]] is a [[Greenhouse Gas]] contributing to [[Global Warming]]. | : [[Methane]] is a [[Greenhouse Gas]] contributing to [[Global Warming]]. | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/0008158770/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158770&linkCode=as2&tag=nrjc-21&linkId=ec31595e720e1529e49876c3866fff6e ''Methane, page 235, GCSE Physics; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782946381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782946381&linkCode=as2&tag=nrjc-21&linkId=5ec5fc3f6429e30c1d9ab9bca2bccf93 ''Methane, page 278, GCSE Combined Science Trilogy; Biology, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945954/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945954&linkCode=as2&tag=nrjc-21&linkId=100574c08fbbb64318256eb79ed61a76 ''Methane, page 343, GCSE Biology, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851362/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851362&linkCode=as2&tag=nrjc-21&linkId=7d78d70a2044ee9982dae010c94af92a ''Methane, pages 109-10, GCSE Combined Science Trilogy 2, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Methane, pages 188, 209, GCSE Combined Science Trilogy; Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Methane, pages 220, 271, GCSE Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359373/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359373&linkCode=as2&tag=nrjc-21&linkId=952a73bbb09d222ecc4b50d200679849 ''Methane, pages 283, 294, GCSE Biology; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Methane, pages 44, 46, 194, 197, 198, 201, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Methane, pages 64-5, 73, 90, 180, 228-9, 234-5, 254, 256, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Methane, pages 21, 36, 161, 231, 256, 257, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
Latest revision as of 10:37, 14 December 2019
Contents
Key Stage 3
Meaning
Methane is a gaseous (at room temperature) hydrocarbon with chemical formula CH4.
About Methane
- Methane is hydrocarbon because it contains only Hydrogen and Carbon atoms.
- Methane can be oxidised to produce Carbon Dioxide and Water.
- Methane + Oxygen → Carbon Dioxide + Water
- Methane is a Greenhouse Gas contributing to Global Warming.
Key Stage 4
Meaning
Methane is a gaseous (at room temperature) alkane with chemical formula CH4.
About Methane
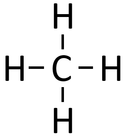
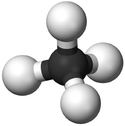
| Chemical Formula (CnH2n+2) | Structural Formula | Structural Diagram | Ball and Stick Model |
| CH4 | CH4 |
- Methane is hydrocarbon because it contains only Hydrogen and Carbon atoms.
- Methane can be oxidised to produce Carbon Dioxide and Water.
- Methane + Oxygen → Carbon Dioxide + Water
- CH4 + 2O2 → CO2 + 2H2O
- Methane is a Greenhouse Gas contributing to Global Warming.
References
AQA
- Methane, page 235, GCSE Physics; Student Book, Collins, AQA
- Methane, page 278, GCSE Combined Science Trilogy; Biology, CGP, AQA
- Methane, page 343, GCSE Biology, CGP, AQA
- Methane, pages 109-10, GCSE Combined Science Trilogy 2, Hodder, AQA
- Methane, pages 188, 209, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Methane, pages 220, 271, GCSE Chemistry, CGP, AQA
- Methane, pages 283, 294, GCSE Biology; Third Edition, Oxford University Press, AQA
- Methane, pages 44, 46, 194, 197, 198, 201, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Methane, pages 64-5, 73, 90, 180, 228-9, 234-5, 254, 256, GCSE Chemistry; Student Book, Collins, AQA