Key Stage 3
Meaning
An atom is a very small particle made of protons, neutrons and electrons that can join with other atoms to make molecules and is considered the smallest part of an element.
- Singular Noun: Atom
- Plural Noun: Atoms
- Adjective: Atomic
About Atoms in The Dalton Model
- In The Dalton Model atoms are shown as ball shaped particles. This makes it easier to draw diagrams of molecules.
About Atoms beyond The Dalton Model
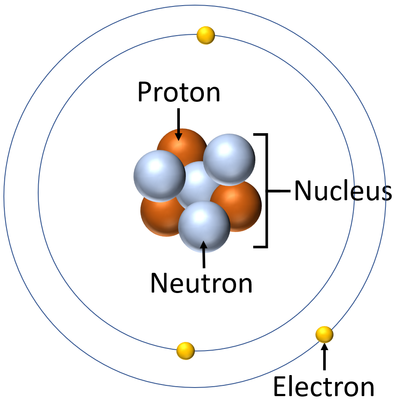
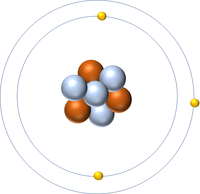
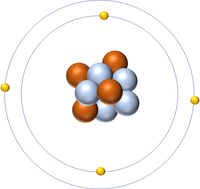
- Atoms are made of three smaller particles; the proton, neutron and electron.
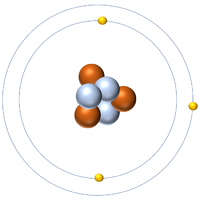
- Protons and neutrons are found in the nucleus at the centre of an atom. Electrons are found orbiting the nucleus in 'shells'.
- In an atom the number of electrons is always the same as the number of protons in the nucleus.
- Different atoms can have different numbers of protons and neutrons.
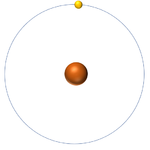
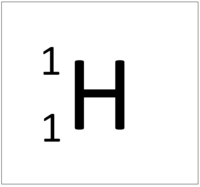
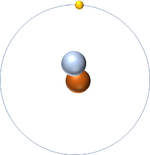
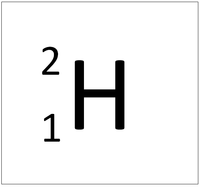
- The simplest atom is Hydrogen which has 1 proton and 1 electron and no neutrons.
Key Stage 4
Meaning
An atom is a very small particle made of protons, neutrons and electrons that can join with other atoms to make molecules and is considered the smallest part of an element.
About Atoms
- Atoms consist of a small, central nucleus containing protons and neutrons surrounded by electrons orbiting the nucleus.
- The electrons orbit the nucleus in so called 'electron shells'.
- Atoms range in size from a diameter of around 1x10-10m (0.1 nanometres) to 5x10-10m (0.5 nanometres), while the nucleus has a diameter of around 1.75x10-15m (1.75 femtometres) to 15x10-15m (15 femtometres). This means the nucleus is 100,000 times smaller than the atom.
- Since molecules are made of multiple atoms they can range from 0.2 nanometres to several millimetres long for long polymer chains.
- In an atom there is always the same number of protons as electrons. If any electron is added or removed the atom becomes an ion.
- Atoms can be different elements depending on the number of protons.
- Atoms of the same element can have different numbers of neutrons so they can be different isotopes of the same element.
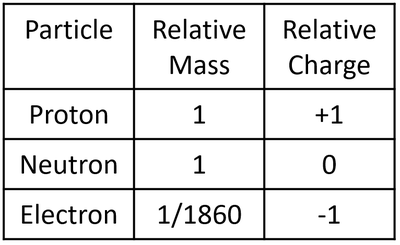
Properties of the Subatomic Particles
- The particles that make an atom all have slightly different properties. Since the particles are so small their mass and charge are extremely small numbers so, to make it easier, they are represented as 'relative' mass and 'relative' charge compared to a proton.
History of Atoms
Discovery of Atoms
- The existence and structure of atoms was not always known.
- An ancient Greek philosopher called Democratus first proposed that matter was made of atoms. He reasoned that if you keep cutting something in half eventually you will reach part of it which cannot be cut into smaller piece. The ancient Greek word for 'uncuttable' was atom.
Dalton Model of the Atom
- It wasn't until the early 1800s that there was an evidence based theory of the existence of an atom. It was proposed by John Dalton who suggested atoms were small spherical objects.
- John Dalton had his theory backed up by the discoveries of Robert Brown who discovered Brownian Motion showing that small invisible particles were responsible for the apparent random motion of small visible particles such as pollen.
Plum Pudding Model of the Atom
- In 1897 J.J. Thompson discovered that there was a particle smaller than an atom which he named the electron after another scientist had hypothesised their existence. The electron was found to be around 2000 times less massive than a Hydrogen atom. It was later realised that these electrons were responsible for electrical current in metals.
- This led him, in 1904, to propose the Plum Pudding Model of the atom in which atoms were believed to be a solid ball of positive charge with electrons stuck inside to give an overall neutral charge to the atom.
Nuclear Model of the Atom
- In 1909 Ernest Rutherford set two students to work on an experiment to probe the structure of the atom in the hope to determine if the Plum Pudding Model was correct.
- Rutherford's students Ernest Marsden and Hans Geiger fired alpha particles, which are positively charged, at a very thin sheet of Gold foil to observe how the alpha particles changed direction as they went through the foil. This was known as Rutherford's Alpha Scattering Experiment.
- They discovered that most of the alpha particles went through in a straight line. A significant number were deflected and a very small number bounced off the Gold back towards the alpha source.
- If the Plum Pudding Model were correct then nearly all of the alpha particles should have passed straight through, unaffected, since the alpha particle is positively charged while atoms should have an even spread of charged particles all the way through them. There should have been no electrostatic force to change their direction.
- This showed that the atom must be mostly empty space, that most of the mass of an atom is concentrated in the centre and that the centre is positively charged. This gave Rutherford evidence to develop a new model of the atom which he proposed in 1911 called the Nuclear Model in which a very small positively charged nucleus is surrounded by orbiting electrons.
The Bohr Model of the Atom
- In 1913 Neils Bohr added to the Nuclear Model when he proposed that electrons could only orbit the nucleus in specific electron orbitals or 'shells' creating the Bohr Model of the atom.
The Addition of the Proton
- In 1919 Ernest Rutherford determined that the Hydrogen nucleus was present in all atomic nuclei. This introduced the proton to the model of the atom.
The Addition of the Neutron
- In 1932 James Chadwick discovered a neutral subatomic particle which he named a Neutron. This neutron explained the existence of isotopes and completed the model of the atom studied at GCSE.
The Electron Cloud
- NB: You don't need to know this The model of the atom was soon updated by Werner Heisenberg who determined that the exact position and velocity of electrons could not be known and so they do not 'orbit', rather they exist in a cloud of probable locations around the nucleus.
References
AQA
- Atom, pages 12-4, 16-7, 20-25, 72, 106, GCSE Chemistry; Student Book, Collins, AQA
- Atom, pages 90, 100, 108-9, 132-3, GCSE Physics; Student Book, Collins, AQA
- Atomic models, page 43, GCSE Physics; The Revision Guide, CGP, AQA
- Atomic models, pages 96, 104, 197, GCSE Combined Science; The Revision Guide, CGP, AQA
- Atomic structure, page 2-3, GCSE Chemistry, Hodder, AQA
- Atomic structure, pages 117-18, 338, GCSE Combined Science Trilogy 1, Hodder, AQA
- Atomic structure, pages 4-21, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Atomic structure; bohr model, page 342, GCSE Combined Science Trilogy 1, Hodder, AQA
- Atomic structure; development of ideas about, pages 123-4, GCSE Combined Science Trilogy 1, Hodder, AQA
- Atomic structure; development of ideas, pages 7-8, GCSE Chemistry, Hodder, AQA
- Atomic structure; electronic structure, page 121, GCSE Combined Science Trilogy 1, Hodder, AQA
- Atoms, page 88, GCSE Physics, Hodder, AQA
- Atoms, pages 12-14, 19, GCSE Chemistry; The Revision Guide, CGP, AQA
- Atoms, pages 22-25, 42-45, 70, 71, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Atoms, pages 22-25, 42-45, 72, 73, GCSE Chemistry, CGP, AQA
- Atoms, pages 4-5, 12-19, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Atoms, pages 50, 92-101, 104-109, GCSE Physics; Third Edition, Oxford University Press, AQA
- Atoms, pages 96-98, 104, 197-199, GCSE Combined Science; The Revision Guide, CGP, AQA
- Atoms; and ions, page 89, GCSE Physics, Hodder, AQA
- Atoms; Bohr model of, page 92, GCSE Physics, Hodder, AQA
- Atoms; discovery of the nucleus, pages 91-2, GCSE Physics, Hodder, AQA
- Atoms; energy levels, page 123, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Atoms; energy levels, pages 111, 201, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Atoms; history, pages 108, 109, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Atoms; history, pages 120, 121, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Atoms; isotopes, page 90, GCSE Physics, Hodder, AQA
- Atoms; nuclear model of, pages 91-2, GCSE Physics, Hodder, AQA
- Atoms; plum-pudding model of, pages 85, 91, GCSE Physics, Hodder, AQA
- Atoms; producing EM radiation, page 201, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Atoms; producing EM radiation, page 243, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Atoms; structure, pages 110, 111, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Atoms; structure, pages 122, 123, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
Edexcel
- Atomic models, pages 354-355, GCSE Combined Science, Pearson Edexcel
- Atomic models, pages 90-91, GCSE Physics, Pearson Edexcel
- Atomic structure, pages 15-17, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Atomic structure, pages 162-163, GCSE Combined Science, Pearson Edexcel
- Atomic structure, pages 18-19, GCSE Chemistry, Pearson, Edexcel
- Atomic structure, pages 32-35, 42, 43, GCSE Chemistry, CGP, Edexcel
- Atomic structure, pages 78-80, 172, 173, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Atomic; models, pages 149-151, GCSE Physics, CGP, Edexcel
- Atoms, page 140, GCSE Physics, Pearson Edexcel
- Atoms, pages 146, 162, 380, GCSE Combined Science, Pearson Edexcel
- Atoms, pages 2, 18, GCSE Chemistry, Pearson, Edexcel
- Atoms, pages 49, 50, GCSE Physics; The Revision Guide, CGP, Edexcel
- Atoms; electromagnetic radiation, pages 351, 354, GCSE Combined Science, Pearson Edexcel
- Atoms; electromagnetic radiation, pages 87, 90, GCSE Physics, Pearson Edexcel
- Atoms; structure, pages 356-357, GCSE Combined Science, Pearson Edexcel
- Atoms; structure, pages 92-93, GCSE Physics, Pearson Edexcel
OCR
- Atoms, Gateway GCSE Physics, Oxford, OCR
- Atoms, pages 13-15, GCSE Chemistry; The Revision Guide, CGP, OCR Gateway
- Atoms, pages 22, 26, 96-97, Gateway GCSE Chemistry, Oxford, OCR
- Atoms; Energy levels, pages 178-179, Gateway GCSE Physics, Oxford, OCR
- Atoms; Isotopes, pages 170-171, Gateway GCSE Physics, Oxford, OCR
- Atoms; Models of, pages 18-21, Gateway GCSE Physics, Oxford, OCR
Beyond the Curriculum