Difference between revisions of "Gas Pressure"
| (8 intermediate revisions by 2 users not shown) | |||
| Line 4: | Line 4: | ||
===About Gas Pressure=== | ===About Gas Pressure=== | ||
| − | : '''Pressure in gases''' is caused by | + | : '''Pressure in gases''' is caused by [[particle]]s [[Collide|colliding]] with the [[object]] or walls of the container. |
| + | : Each time a [[particle]] in the [[gas]] hits an [[object]] or the walls it provides a [[force]]. | ||
| + | : The more [[particle]]s that hit the [[object]] or walls, the bigger the overall [[force]] it will experience. | ||
| + | : The faster the [[particle]]s hit the [[object]] or walls, the bigger the [[force]] that each [[particle]] [[collide]]s with. | ||
| + | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| Line 21: | Line 25: | ||
| style="height:20px; width:200px; text-align:center;" |Increasing the number of particles in the container. | | style="height:20px; width:200px; text-align:center;" |Increasing the number of particles in the container. | ||
|} | |} | ||
| + | |||
| + | ===Extra Information=== | ||
| + | {{#ev:youtube|https://www.youtube.com/watch?v=VDf00z8sMFw}} | ||
| + | |||
| + | ==Key Stage 4== | ||
| + | ===Meaning=== | ||
| + | '''Gas pressure''' is the [[pressure]] on an [[object]] caused by a [[gas]]. | ||
| + | |||
| + | ===About Gas Pressure=== | ||
| + | : '''Pressure in gases''' is caused by [[particle]]s [[Collide|colliding]] with the [[object]] or walls of the container. | ||
| + | : The [[pressure]] from a [[gas]] always acts at [[Right Angle|right angles]] to the surface. | ||
| + | : Each time a [[particle]] in the [[gas]] hits an [[object]] or the walls it changes [[momentum]] which causes a [[force]] to be applied to the surface. | ||
| + | : The more [[particle]]s that hit the surface, the bigger the overall [[force]] it will experience. | ||
| + | : The faster the [[particle]]s hit the [[object]] or walls, the bigger the [[force]] that each [[particle]] [[collide]]s with. | ||
| + | : The frequency and [[speed]] with which the [[particle]]s collide with the surface determines the [[pressure]] on that surface. | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | |[[File:GasPressure1.png|center|300px]] | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |The [[Particle Model|particle model]] of a [[gas]] showing the [[particle]]s as red balls and their speed and direction shown by the arrows. The longer the arrows the faster they are moving. | ||
| + | |} | ||
| + | |||
| + | : The '''gas pressure''' can be increased in three ways: | ||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | |[[File:GasPressure3.png|center|200px]] | ||
| + | |[[File:GasPressure2.png|center|200px]] | ||
| + | |[[File:GasPressure4.png|center|200px]] | ||
| + | |- | ||
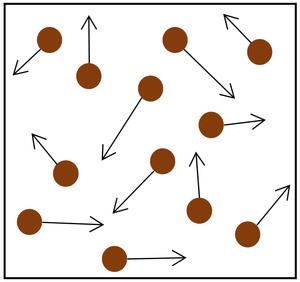
| + | | style="height:20px; width:200px; text-align:center;" |Increasing the [[temperature]] of the [[gas]] which makes the [[particle]]s move faster. This causes the particles to hit the walls of the container more often and with a greater [[force]]. | ||
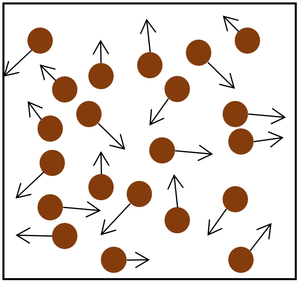
| + | | style="height:20px; width:200px; text-align:center;" |Increasing the number of [[particle]]s in the container. | ||
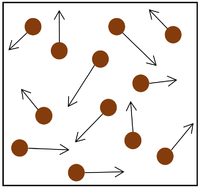
| + | | style="height:20px; width:200px; text-align:center;" |Decreasing the [[Volume (Space)|volume]] of the container. The additional [[pressure]] caused by the [[force]] causes the [[gas]] to be [[compression|compressed]]. The [[force]] does [[Work Done|work]] on the [[gas]] increasing its [[temperature]]. | ||
| + | |} | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Gas pressure, pages 330-1, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851370/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851370&linkCode=as2&tag=nrjc-21&linkId=01c69b0ae058f809cf636033e6ba793e ''Gas pressure, pages 78-9, GCSE Physics, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/019835939X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=019835939X&linkCode=as2&tag=nrjc-21&linkId=57e96876985fc39b1a3d8a3e3dc238b6 ''Gas pressure, pages 86-89, GCSE Physics; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851370/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851370&linkCode=as2&tag=nrjc-21&linkId=01c69b0ae058f809cf636033e6ba793e ''Gas pressure; demonstration of, page 79, GCSE Physics, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Gas pressure; effect on equilibrium position, pages 162-3, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Gas pressure; effect on rate of reactions, pages 152, 154, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851370/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851370&linkCode=as2&tag=nrjc-21&linkId=01c69b0ae058f809cf636033e6ba793e ''Gas pressure; expanding and compressing gases, pages 79-80, GCSE Physics, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851370/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851370&linkCode=as2&tag=nrjc-21&linkId=01c69b0ae058f809cf636033e6ba793e ''Gas pressure; explanation of, page 80, GCSE Physics, Hodder, AQA ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359837/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359837&linkCode=as2&tag=nrjc-21&linkId=3c4229e8b023b2b60768e7ea2307cc6f ''Gas pressure, page 34-35, 37, Gateway GCSE Physics, Oxford, OCR ''] | ||
Latest revision as of 11:21, 11 December 2019
Contents
Key Stage 3
Meaning
Gas pressure is the pressure on an object caused by a gas.
About Gas Pressure
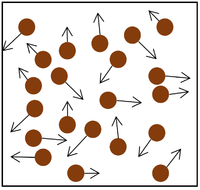
- Pressure in gases is caused by particles colliding with the object or walls of the container.
- Each time a particle in the gas hits an object or the walls it provides a force.
- The more particles that hit the object or walls, the bigger the overall force it will experience.
- The faster the particles hit the object or walls, the bigger the force that each particle collides with.
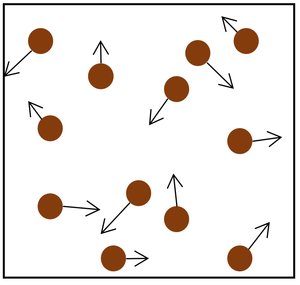
| The particle model of a gas showing the particles as red balls and their speed and direction shown by the arrows. The longer the arrows the faster they are moving. |
- The gas pressure can be increased in two ways:
| Increasing the temperature of the gas which makes the particles move faster. This causes the particles to hit the walls of the container more often and with a greater force. | Increasing the number of particles in the container. |
Extra Information
Key Stage 4
Meaning
Gas pressure is the pressure on an object caused by a gas.
About Gas Pressure
- Pressure in gases is caused by particles colliding with the object or walls of the container.
- The pressure from a gas always acts at right angles to the surface.
- Each time a particle in the gas hits an object or the walls it changes momentum which causes a force to be applied to the surface.
- The more particles that hit the surface, the bigger the overall force it will experience.
- The faster the particles hit the object or walls, the bigger the force that each particle collides with.
- The frequency and speed with which the particles collide with the surface determines the pressure on that surface.
| The particle model of a gas showing the particles as red balls and their speed and direction shown by the arrows. The longer the arrows the faster they are moving. |
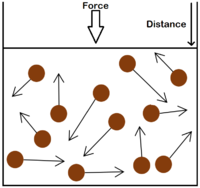
- The gas pressure can be increased in three ways:
| Increasing the temperature of the gas which makes the particles move faster. This causes the particles to hit the walls of the container more often and with a greater force. | Increasing the number of particles in the container. | Decreasing the volume of the container. The additional pressure caused by the force causes the gas to be compressed. The force does work on the gas increasing its temperature. |
References
AQA
- Gas pressure, pages 330-1, GCSE Combined Science Trilogy 1, Hodder, AQA
- Gas pressure, pages 78-9, GCSE Physics, Hodder, AQA
- Gas pressure, pages 86-89, GCSE Physics; Third Edition, Oxford University Press, AQA
- Gas pressure; demonstration of, page 79, GCSE Physics, Hodder, AQA
- Gas pressure; effect on equilibrium position, pages 162-3, GCSE Chemistry, Hodder, AQA
- Gas pressure; effect on rate of reactions, pages 152, 154, GCSE Chemistry, Hodder, AQA
- Gas pressure; expanding and compressing gases, pages 79-80, GCSE Physics, Hodder, AQA
- Gas pressure; explanation of, page 80, GCSE Physics, Hodder, AQA