Difference between revisions of "Gas Pressure"
(→Key Stage 3) |
(→Key Stage 4) |
||
| Line 54: | Line 54: | ||
| style="height:20px; width:200px; text-align:center;" |Increasing the [[temperature]] of the [[gas]] which makes the [[particle]]s move faster. This causes the particles to hit the walls of the container more often and with a greater [[force]]. | | style="height:20px; width:200px; text-align:center;" |Increasing the [[temperature]] of the [[gas]] which makes the [[particle]]s move faster. This causes the particles to hit the walls of the container more often and with a greater [[force]]. | ||
| style="height:20px; width:200px; text-align:center;" |Increasing the number of [[particle]]s in the container. | | style="height:20px; width:200px; text-align:center;" |Increasing the number of [[particle]]s in the container. | ||
| − | | style="height:20px; width:200px; text-align:center;" |Decreasing the [[Volume (Space)|volume]] of the container. | + | | style="height:20px; width:200px; text-align:center;" |Decreasing the [[Volume (Space)|volume]] of the container. The additional [[pressure]] caused by the [[force]] causes the [[gas]] to be [[compression|compressed]]. The [[force]] does [[Work Done|work]] on the [[gas]] increasing its [[temperature]]. |
|} | |} | ||
Revision as of 14:27, 6 March 2019
Contents
Key Stage 3
Meaning
Gas pressure is the pressure on an object caused by a gas.
About Gas Pressure
- Pressure in gases is caused by particles colliding with the object or walls of the container.
- Each time a particle in the gas hits an object or the walls it provides a force.
- The more particles that hit the object or walls, the bigger the overall force it will experience.
- The faster the particles hit the object or walls, the bigger the force that each particle collides with.
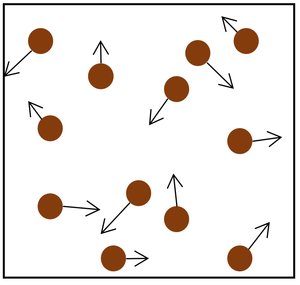
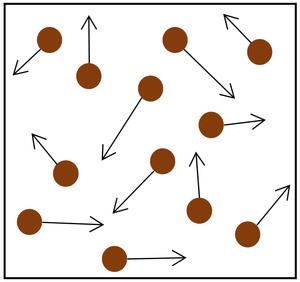
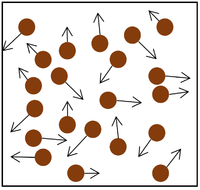
| The particle model of a gas showing the particles as red balls and their speed and direction shown by the arrows. The longer the arrows the faster they are moving. |
- The gas pressure can be increased in two ways:
| Increasing the temperature of the gas which makes the particles move faster. This causes the particles to hit the walls of the container more often and with a greater force. | Increasing the number of particles in the container. |
Key Stage 4
Meaning
Gas pressure is the pressure on an object caused by a gas.
About Gas Pressure
- Pressure in gases is caused by particles colliding with the object or walls of the container.
- The pressure from a gas always acts at right angles to the surface.
- Each time a particle in the gas hits an object or the walls it changes momentum which causes a force to be applied to the surface.
- The more particles that hit the surface, the bigger the overall force it will experience.
- The faster the particles hit the object or walls, the bigger the force that each particle collides with.
- The frequency and speed with which the particles collide with the surface determines the pressure on that surface.
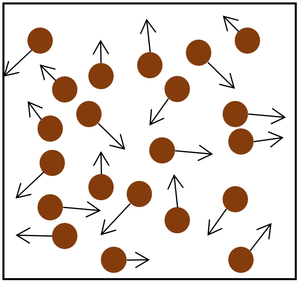
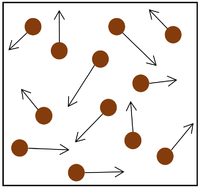
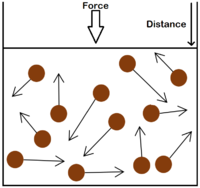
| The particle model of a gas showing the particles as red balls and their speed and direction shown by the arrows. The longer the arrows the faster they are moving. |
- The gas pressure can be increased in three ways:
| Increasing the temperature of the gas which makes the particles move faster. This causes the particles to hit the walls of the container more often and with a greater force. | Increasing the number of particles in the container. | Decreasing the volume of the container. The additional pressure caused by the force causes the gas to be compressed. The force does work on the gas increasing its temperature. |