Difference between revisions of "Nuclear Fusion"
| Line 6: | Line 6: | ||
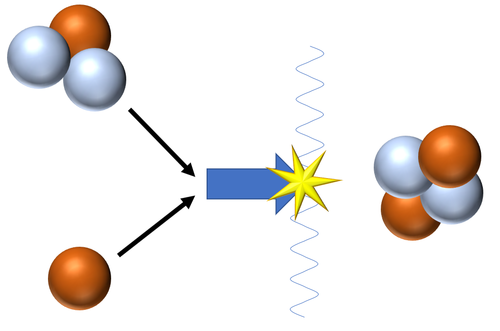
: In a '''nuclear fusion reaction''' the products have less [[mass]] than the reactants as some of the [[Rest Mass Energy|rest mass]] is converted into [[energy]] in the process. | : In a '''nuclear fusion reaction''' the products have less [[mass]] than the reactants as some of the [[Rest Mass Energy|rest mass]] is converted into [[energy]] in the process. | ||
: For a '''fusion''' [[Nuclear Reaction|reaction]] to occur the two [[Atomic Nucleus|nuclei]] must have enough [[Kinetic Energy Store|kinetic energy]] to overcome the [[Electrostatic Force|electrostatic force]] of repulsion between the [[Positive Charge|positively charged]] [[Atomic Nucleus|nuclei]]. | : For a '''fusion''' [[Nuclear Reaction|reaction]] to occur the two [[Atomic Nucleus|nuclei]] must have enough [[Kinetic Energy Store|kinetic energy]] to overcome the [[Electrostatic Force|electrostatic force]] of repulsion between the [[Positive Charge|positively charged]] [[Atomic Nucleus|nuclei]]. | ||
| − | : To provide enough [[Kinetic Energy Store|kinetic energy]] to the [[Atomic | + | : To provide enough [[Kinetic Energy Store|kinetic energy]] to the [[Atomic Nucleus|nuclei]] the [[substance]] must be [[heat]]ed to a [[temperature]] of several million [[Degrees Celsius|degrees Celsius]]. |
: '''Nuclear fusion''' occurs naturally in the centre of a [[Star]] due to the high [[temperature]]s and [[pressure]]. | : '''Nuclear fusion''' occurs naturally in the centre of a [[Star]] due to the high [[temperature]]s and [[pressure]]. | ||
: '''Nuclear fusion''' is possible in [[laboratory|laboratories]] on [[Earth]] but it cannot be sustained for long periods of [[time]] to produce enough [[energy]] to be useful as an [[Energy Resource|energy resource]]. | : '''Nuclear fusion''' is possible in [[laboratory|laboratories]] on [[Earth]] but it cannot be sustained for long periods of [[time]] to produce enough [[energy]] to be useful as an [[Energy Resource|energy resource]]. | ||
Revision as of 08:27, 8 April 2019
Key Stage 4
Meaning
Nuclear fusion is a process in which two small atomic nuclei join together to become a larger nucleus.
About Nuclear Fusion
- In a nuclear fusion reaction the products have less mass than the reactants as some of the rest mass is converted into energy in the process.
- For a fusion reaction to occur the two nuclei must have enough kinetic energy to overcome the electrostatic force of repulsion between the positively charged nuclei.
- To provide enough kinetic energy to the nuclei the substance must be heated to a temperature of several million degrees Celsius.
- Nuclear fusion occurs naturally in the centre of a Star due to the high temperatures and pressure.
- Nuclear fusion is possible in laboratories on Earth but it cannot be sustained for long periods of time to produce enough energy to be useful as an energy resource.
- Nuclear fusion in laboratories on Earth must be done at much higher temperature than in the centre of Stars because the centre of Stars is a much higher pressure so nuclei collide more often.
Nuclear Fusion Reactions
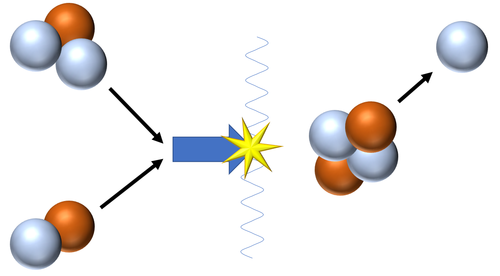
| Fusion of a Tritium and a Deuterium nucleus produces a Helium nucleus.
\({}_1^2H + {}_1^3H \rightarrow {}_2^4He + {}_0^1n\) This is the most common pathway for nuclear fusion in Stars as the neutron goes on to be captured by a Hydrogen nucleus to become Deuterium or captured by a Deuterium nucleus to become a Tritium nucleus. |
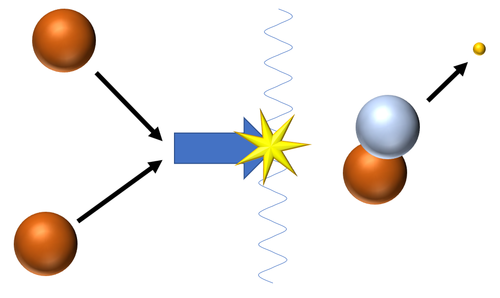
| Fusion of two Hydrogen nuclei (protons) produces a Deuterium nucleus.
\({}_1^1H + {}_1^1H \rightarrow {}_1^2H + {}_{-1}^0\beta\) |
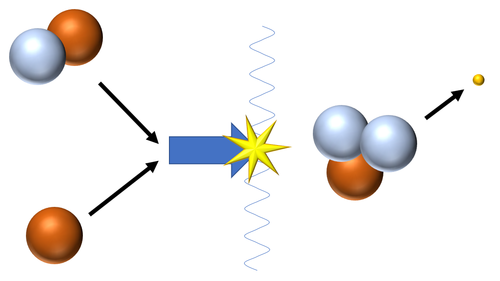
| Fusion of a Hydrogen nucleus (proton) and a Deuterium nucleus produces a Tritium nucleus.
\({}_1^1H + {}_1^2H \rightarrow {}_1^3H + {}_{-1}^0\beta\) |
| Fusion of two Deuterium nuclei produces a Helium nucleus.
\({}_1^2H + {}_1^2H \rightarrow {}_2^4He\) |
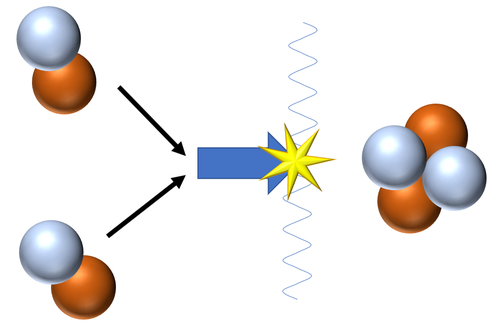
| Fusion of a Hydrogen nucleus (proton) and a Tritium nucleus produces a Helium nucleus.
\({}_1^1H + {}_1^3H \rightarrow {}_2^4He\) |