Difference between revisions of "Indicator (Chemistry)"
(→About Indicators) |
|||
| Line 62: | Line 62: | ||
|- | |- | ||
|} | |} | ||
| + | |||
| + | ===References=== | ||
| + | ====Edexcel==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Indicators (pH) page 52, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Indicators (pH), page 196, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Indicators, page 43, 65, 110, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Indicators, pages 105, 209, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Indicators, pages 118-120, 131, 132, 185, 186, 205, 323, GCSE Chemistry, CGP, Edexcel ''] | ||
Revision as of 15:38, 21 November 2019
Contents
Key Stage 3
Meaning
An indicator is a dye that changes colour when other chemicals are present.
About Indicators
Some non-pH indicators you should know:
- Iodine Solution - Used to detect starch.
- Biuret Solution - Used to detect proteins.
- Benedict's Solution - Used to detect simple sugars like glucose.
- Limewater - Used to detect Carbon Dioxide.
About pH Indicators
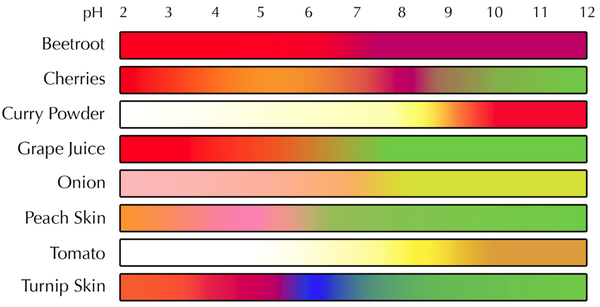
- The colour of a pH indicator can be used to tell the pH of a solution.
- Different indicators will have a different range of colours for different pH values.
- A good indicator can be added to solution without affecting the pH of the solution. If an indicator change the pH of a solution it could not give an accurate reading.
Some pH indicators you should know:
- Litmus Paper
- Red Cabbage Indicator
- Universal Indicator
- Phenolphthalein
- Methyl Orange
- Bromothymol Blue
Examples
Key Stage 4
Meaning
An indicator is a dye that changes colour when other chemicals are present.
About Indicators
Some non-pH indicators you should know:
- Iodine Solution - Used to detect starch.
- Biuret Solution - Used to detect proteins.
- Benedict's Solution - Used to detect simple sugars like glucose.
- Limewater - Used to detect Carbon Dioxide.
About pH Indicators
- The colour of a pH indicator can be used to tell the pH of a solution.
- Different indicators will have a different range of colours for different pH values.
- A good indicator can be added to solution without affecting the pH of the solution. If an indicator change the pH of a solution it could not give an accurate reading.
Some pH indicators you should know:
- Litmus Paper
- Red Cabbage Indicator
- Universal Indicator
- Phenolphthalein
- Methyl Orange
- Bromothymol Blue
Examples
References
Edexcel
- Indicators (pH) page 52, GCSE Chemistry, Pearson, Edexcel
- Indicators (pH), page 196, GCSE Combined Science, Pearson Edexcel
- Indicators, page 43, 65, 110, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Indicators, pages 105, 209, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Indicators, pages 118-120, 131, 132, 185, 186, 205, 323, GCSE Chemistry, CGP, Edexcel