Contents
Key Stage 3
Meaning
An indicator is a dye that changes colour when other chemicals are present.
About Indicators
- Some indicators detect pH but others can be used to detect other chemicals.
Some non-pH indicators you should know:
- Iodine Solution - Used to detect starch.
- Biuret Solution - Used to detect proteins.
- Benedict's Solution - Used to detect simple sugars like glucose.
- Limewater - Used to detect Carbon Dioxide.
About pH Indicators
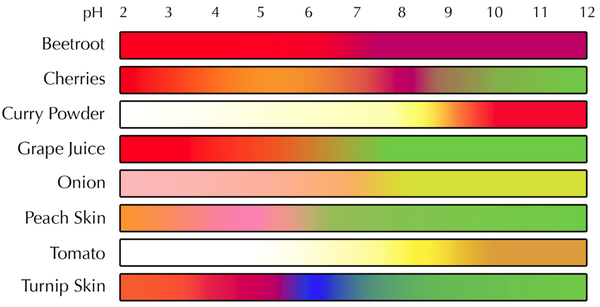
- The colour of a pH indicator can be used to tell the pH of a solution.
- Different indicators will have a different range of colours for different pH values.
- A good indicator can be added to solution without affecting the pH of the solution. If an indicator change the pH of a solution it could not give an accurate reading.
Some pH indicators you should know:
- Litmus Paper
- Red Cabbage Indicator
- Universal Indicator
- Phenolphthalein
- Methyl Orange
- Bromothymol Blue
Examples
Key Stage 4
Meaning
An indicator is a dye that changes colour when other chemicals are present.
About Indicators
- Some indicators detect pH but others can be used to detect other chemicals.
Some non-pH indicators you should know:
- Iodine Solution - Used to detect starch.
- Biuret Solution - Used to detect proteins.
- Benedict's Solution - Used to detect simple sugars like glucose.
- Limewater - Used to detect Carbon Dioxide.
About pH Indicators
- The colour of a pH indicator can be used to tell the pH of a solution.
- Different indicators will have a different range of colours for different pH values.
- A good indicator can be added to solution without affecting the pH of the solution. If an indicator change the pH of a solution it could not give an accurate reading.
Some pH indicators you should know:
- Litmus Paper
- Red Cabbage Indicator
- Universal Indicator
- Phenolphthalein
- Methyl Orange
- Bromothymol Blue