Difference between revisions of "Ethane"
| Line 43: | Line 43: | ||
:[https://www.amazon.co.uk/gp/product/1292120207/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120207&linkCode=as2&tag=nrjc-21&linkId=22455ff53961978667722edaa64c0be5 ''Ethane gas, pages 137, 139, GCSE Biology, Pearson, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120207/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120207&linkCode=as2&tag=nrjc-21&linkId=22455ff53961978667722edaa64c0be5 ''Ethane gas, pages 137, 139, GCSE Biology, Pearson, Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1782948120/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948120&linkCode=as2&tag=nrjc-21&linkId=dedef775c6a43dbb0a609441525adac0 ''Ethane, page 219, GCSE Biology, CGP, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1782948120/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948120&linkCode=as2&tag=nrjc-21&linkId=dedef775c6a43dbb0a609441525adac0 ''Ethane, page 219, GCSE Biology, CGP, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945660/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945660&linkCode=as2&tag=nrjc-21&linkId=83aa4500ad7759e7f401a1c5ba5df758 ''Ethane, page 51, Gateway GCSE Biology; The Revision Guide, CGP, OCR ''] | ||
Latest revision as of 15:13, 6 December 2019
Contents
Key Stage 3
Meaning
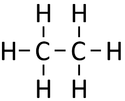
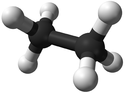
Ethane is a gaseous (at room temperature) hydrocarbon with chemical formula C2H6.
About Ethane
- Ethane is hydrocarbon because it contains only Hydrogen and Carbon atoms.
- Ethane can be oxidised to produce Carbon Dioxide and Water.
- Ethane + Oxygen → Carbon Dioxide + Water
Key Stage 4
Meaning
Ethane is a gaseous (at room temperature) alkane with chemical formula C2H6.
About Ethane
| Chemical Formula (CnH2n+2) | Structural Formula | Structural Diagram | Ball and Stick Model |
| C2H6 | CH3CH3 |
- Ethane is hydrocarbon because it contains only Hydrogen and Carbon atoms.
- Ethane can be oxidised to produce Carbon Dioxide and Water.
- Ethane + Oxygen → Carbon Dioxide + Water
- 2C2H6 + 7O2 → 4CO2 + 6H2O
References
AQA
- Ethane, page 188, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Ethane, page 195, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Ethane, page 220, GCSE Chemistry, CGP, AQA
- Ethane, pages 99, 228-9, 238, 254, 256, GCSE Chemistry; Student Book, Collins, AQA
Edexcel
- Ethane gas, pages 137, 139, GCSE Biology, Pearson, Edexcel
- Ethane, page 219, GCSE Biology, CGP, Edexcel