Difference between revisions of "Lattice"
| Line 33: | Line 33: | ||
:[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Lattices; ionic, pages 180-181, GCSE Combined Science, Pearson Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Lattices; ionic, pages 180-181, GCSE Combined Science, Pearson Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Lattices; ionic, pages 36-37, GCSE Chemistry, Pearson, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Lattices; ionic, pages 36-37, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Lattice structures, page 19, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Lattice structures, page 91, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Lattice structures, pages 59, 62-63, 66, 78, 86, 220, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
Latest revision as of 16:28, 13 December 2019
Contents
Key Stage 4
Meaning
A lattice is a structure made of a regularly repeating pattern of atoms.
About Lattices
Examples
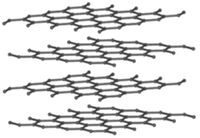
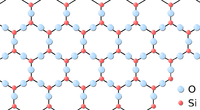
| Graphite is a lattice of Carbon atoms forming many layers which join together. | Graphene is a hexagonal lattice of Carbon atoms making a single layer. | Silica is a hexagonal lattice made of Silicon and Oxygen atoms. |
References
AQA
- Lattices, pages 42-43, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Lattices, pages 79, 90, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Lattices, pages 81, 92, GCSE Chemistry, CGP, AQA
Edexcel
- Lattice structures, pages 22, 24, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Lattice structures, pages 54, 55, GCSE Chemistry, CGP, Edexcel
- Lattices; giant, page 190, GCSE Combined Science, Pearson Edexcel
- Lattices; giant, page 46, GCSE Chemistry, Pearson, Edexcel
- Lattices; ionic, pages 180-181, GCSE Combined Science, Pearson Edexcel
- Lattices; ionic, pages 36-37, GCSE Chemistry, Pearson, Edexcel