Difference between revisions of "Fuel Cell"
| Line 7: | Line 7: | ||
: In a '''fuel cell''' [[Oxygen]] is combined with [[Hydrogen]] to produce [[Water]]. | : In a '''fuel cell''' [[Oxygen]] is combined with [[Hydrogen]] to produce [[Water]]. | ||
: '''Fuel cells''' are designed to combine [[Hydrogen Ion|Hydrogen ions]] and [[Hydroxide Ion|Hydroxide ions]] to produce a [[Potential Difference|potential difference]] between two [[electrode]]s. | : '''Fuel cells''' are designed to combine [[Hydrogen Ion|Hydrogen ions]] and [[Hydroxide Ion|Hydroxide ions]] to produce a [[Potential Difference|potential difference]] between two [[electrode]]s. | ||
| − | : '''Fuel cells''' may be used in electric cars and were used on the | + | : '''Fuel cells''' may be used in electric cars and were used on the Space Shuttle. |
===Advantages=== | ===Advantages=== | ||
Revision as of 19:48, 3 April 2019
Key Stage 4
Meaning
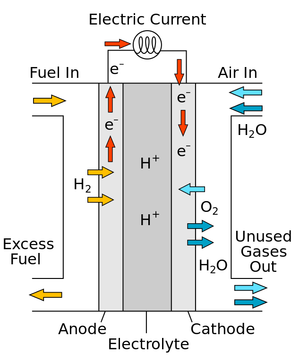
A diagram of a simple fuel cell.
A fuel cell is a device which can combine Hydrogen and Oxygen to produce a Potential Difference.
About Fuel Cells
- In a fuel cell Oxygen is combined with Hydrogen to produce Water.
- Fuel cells are designed to combine Hydrogen ions and Hydroxide ions to produce a potential difference between two electrodes.
- Fuel cells may be used in electric cars and were used on the Space Shuttle.
Advantages
- No Carbon Dioxide is produced.
- Refilling with Hydrogen is quicker than recharging a battery.
- They can be made in many different sizes for different uses.
Disadvantages
- Hydrogen must be stored as a Compressed Gas.
- Hydrogen is highly flammable.
- Hydrogen is made by electrolysis which requires electricity, which is often made by power stations burning fuel and producing Carbon Dioxide.