Difference between revisions of "Diffusion"
(→Key Stage 4) |
|||
| Line 4: | Line 4: | ||
===About Diffusion=== | ===About Diffusion=== | ||
| − | : [[Diffusion]] can only happen in a [[fluid]] | + | : [[Diffusion]] can only happen in a [[fluid]] (a [[liquid]] or a [[gas]]) because the [[particle]]s can move past each other. |
: [[Diffusion]] cannot happen in a [[solid]] because the [[particle]]s are held in fixed positions. | : [[Diffusion]] cannot happen in a [[solid]] because the [[particle]]s are held in fixed positions. | ||
| − | : In [[diffusion]] the | + | : In [[diffusion]] the [[particle]]s always spread from a high [[concentration]] (where there are lots of the [[particle]]s, to a low [[concentration]] (where there are less of the [[particle]]s). |
| − | |||
{| class="wikitable" | {| class="wikitable" | ||
| Line 25: | Line 24: | ||
==Key Stage 4== | ==Key Stage 4== | ||
===Meaning=== | ===Meaning=== | ||
| − | [[Diffusion]] is when [[ | + | [[Diffusion]] is when [[particle]]s spread from a region of high [[concentration]] to a region of low [[concentration]] along a [[Concentration Gradient|concentration gradient]]. |
| + | |||
| + | ===About Diffusion=== | ||
| + | : [[Diffusion]] can only happen in a [[fluid]] (a [[liquid]] or a [[gas]]) because the [[particle]]s can move past each other. | ||
| + | : [[Diffusion]] cannot happen in a [[solid]] because the [[particle]]s are held in fixed positions. | ||
| + | : In [[diffusion]] the [[particle]]s always follow the [[Concentration Gradient|concentration gradient]] spreading from a high [[concentration]] (where there is lots of the [[particle]]s), to a low [[concentration]] (where there are less of the [[particle]]s). | ||
| + | : [[Diffusion]] stops when all [[substance]]s are spread out equally and there is no longer a [[Concentration Gradient|concentration gradient]]. | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | |[[File:Diffusion.png|center|600px]] | ||
| + | |- | ||
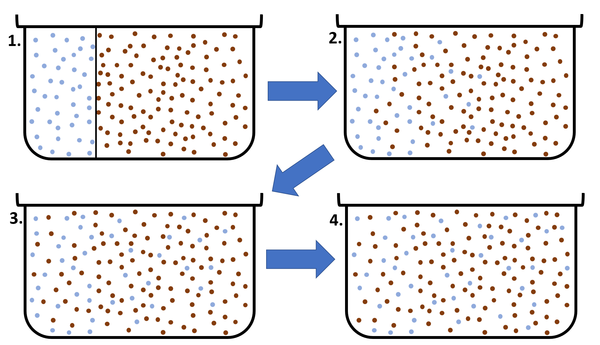
| + | | style="height:20px; width:600px; text-align:center;" |This [[diagram]] shows a high [[concentration]] of blue [[particle]]s on the left separated from the red [[particle]]s by a barrier. When the barrier is removed the blue [[particle]]s [[Diffusion|diffuse]] to the area of lower [[concentration]] on the right. [[Diffusion]] continues until all [[particle]]s are equally spread. | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | |[[File:Diffusion.gif|center]] | ||
| + | |- | ||
| + | | style="height:20px; width:600px; text-align:center;" |This animation shows a high [[concentration]] of red [[particle]]s initially at the top. When [[diffusion]] begins the red [[particle]]s spread to the area of lower [[concentration]] at the bottom. [[Diffusion]] continues until all [[particle]]s are equally spread. | ||
| + | |} | ||
Revision as of 08:48, 5 June 2019
Contents
Key Stage 3
Meaning
Diffusion is when particles spread from a region of high concentration to a region of low concentration.
About Diffusion
- Diffusion can only happen in a fluid (a liquid or a gas) because the particles can move past each other.
- Diffusion cannot happen in a solid because the particles are held in fixed positions.
- In diffusion the particles always spread from a high concentration (where there are lots of the particles, to a low concentration (where there are less of the particles).
| This diagram shows a high concentration of blue particles on the left separated from the red particles by a barrier. When the barrier is removed the blue particles diffuse to the area of lower concentration on the right. Diffusion continues until all particles are equally spread. |
| This animation shows a high concentration of red particles initially at the top. When diffusion begins the red particles spread to the area of lower concentration at the bottom. Diffusion continues until all particles are equally spread. |
Key Stage 4
Meaning
Diffusion is when particles spread from a region of high concentration to a region of low concentration along a concentration gradient.
About Diffusion
- Diffusion can only happen in a fluid (a liquid or a gas) because the particles can move past each other.
- Diffusion cannot happen in a solid because the particles are held in fixed positions.
- In diffusion the particles always follow the concentration gradient spreading from a high concentration (where there is lots of the particles), to a low concentration (where there are less of the particles).
- Diffusion stops when all substances are spread out equally and there is no longer a concentration gradient.
| This diagram shows a high concentration of blue particles on the left separated from the red particles by a barrier. When the barrier is removed the blue particles diffuse to the area of lower concentration on the right. Diffusion continues until all particles are equally spread. |
| This animation shows a high concentration of red particles initially at the top. When diffusion begins the red particles spread to the area of lower concentration at the bottom. Diffusion continues until all particles are equally spread. |