Difference between revisions of "Litmus Paper"
(Created page with "==Key Stage 3== ===Meaning=== right|300px|thumb|Two pieces of '''litmus paper''' dipped in an [[acid showing the red '''litmus''' stayed red but the b...") |
|||
| Line 6: | Line 6: | ||
===About Litmus Paper=== | ===About Litmus Paper=== | ||
: '''Litmus paper''' comes in two colours: Red and Blue. | : '''Litmus paper''' comes in two colours: Red and Blue. | ||
| − | : '''Litmus | + | : '''Litmus paper''' is a very simple [[Indicator (Chemistry|indicator]] as it can only tell if something is [[acid]] or [[alkali]] but it cannot tell the exact [[pH]] of a [[solution]]. |
| − | : ''' | + | : '''Litmus paper''' cannot be used on a [[base]] unless it is in [[solution]]. |
| − | * | + | * |
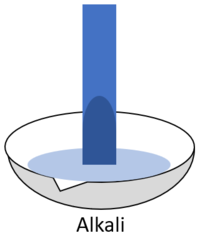
*When red '''litmus''' is placed in an [[alkali]] it turns blue. | *When red '''litmus''' is placed in an [[alkali]] it turns blue. | ||
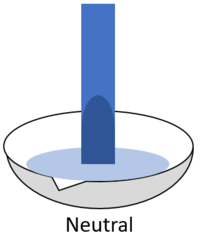
| − | *When red '''litmus''' is placed in a [[neutral]] [[solution]] it stays red. | + | * |
| − | + | * | |
| − | + | * | |
| − | + | * | |
| + | |||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | |[[File:RedLitmusAcid.png|center|200px]] | ||
| + | |[[File:RedLitmusAlkali.png|center|200px]] | ||
| + | |[[File:RedLitmusNeutral.png|center|200px]] | ||
| + | |- | ||
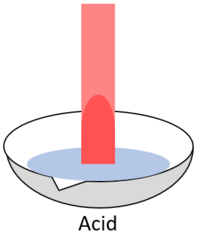
| + | | style="height:20px; width:200px; text-align:center;" |When red '''litmus''' is placed in an [[acid]] it stays red. | ||
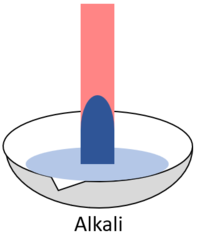
| + | | style="height:20px; width:200px; text-align:center;" |When red '''litmus''' is placed in an [[alkali]] it turns blue. | ||
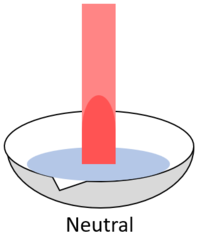
| + | | style="height:20px; width:200px; text-align:center;" |When red '''litmus''' is placed in a [[neutral]] [[solution]] it stays red. | ||
| + | |- | ||
| + | |[[File:BlueLitmusAcid.png|center|200px]] | ||
| + | |[[File:BlueLitmusAlkali.png|center|200px]] | ||
| + | |[[File:BlueLitmusNeutral.png|center|200px]] | ||
| + | |- | ||
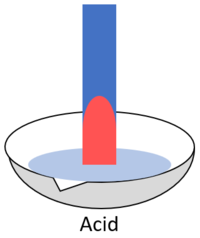
| + | | style="height:20px; width:200px; text-align:center;" |When blue '''litmus''' is placed in an [[acid]] it turns red. | ||
| + | | style="height:20px; width:200px; text-align:center;" |When blue '''litmus''' is placed in an [[alkali]] it stays blue. | ||
| + | | style="height:20px; width:200px; text-align:center;" |When blue '''litmus''' is placed in a [[neutral]] [[solution]] it stays blue. | ||
| + | |} | ||
Revision as of 09:18, 29 September 2018
Key Stage 3
Meaning

Two pieces of litmus paper dipped in an acid showing the red litmus stayed red but the blue litmus turned red.
Litmus paper is a piece of paper coloured with a dye that turns red in acid and blue in alkali.
About Litmus Paper
- Litmus paper comes in two colours: Red and Blue.
- Litmus paper is a very simple indicator as it can only tell if something is acid or alkali but it cannot tell the exact pH of a solution.
- Litmus paper cannot be used on a base unless it is in solution.
- When red litmus is placed in an alkali it turns blue.
| When red litmus is placed in an acid it stays red. | When red litmus is placed in an alkali it turns blue. | When red litmus is placed in a neutral solution it stays red. |
| When blue litmus is placed in an acid it turns red. | When blue litmus is placed in an alkali it stays blue. | When blue litmus is placed in a neutral solution it stays blue. |