Difference between revisions of "Litmus Paper"
| Line 34: | Line 34: | ||
:[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Litmus, page 43, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Litmus, page 43, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Litmus, pages 119, 186, 213, 275, 323, GCSE Chemistry, CGP, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Litmus, pages 119, 186, 213, 275, 323, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Litmus paper, pages 146, 267, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Litmus paper, pages 58, 103, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
Latest revision as of 08:58, 14 December 2019
Key Stage 3
Meaning

Two pieces of litmus paper dipped in an acid showing the red litmus stayed red but the blue litmus turned red.
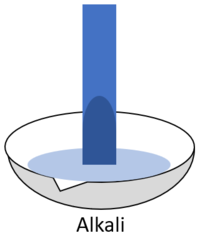
Litmus paper is a piece of paper coloured with a dye that turns red in acid and blue in alkali.
About Litmus Paper
- Litmus paper comes in two colours: Red and Blue.
- Litmus paper is a very simple indicator as it can only tell if something is acid or alkali but it cannot tell the exact pH of a solution.
- Litmus paper cannot be used on a base unless it is in solution.
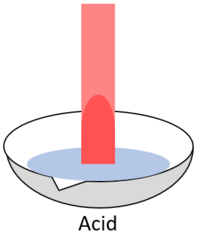
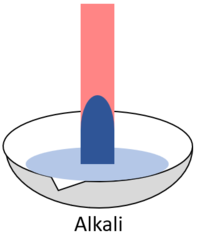
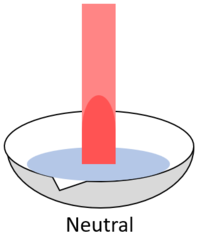
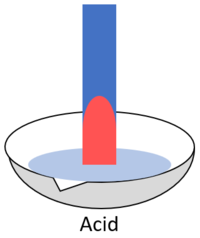
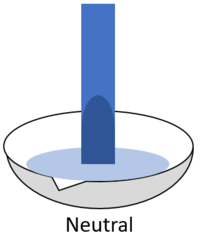
| When red litmus is placed in an acid it stays red. | When red litmus is placed in an alkali it turns blue. | When red litmus is placed in a neutral solution it stays red. |
| When blue litmus is placed in an acid it turns red. | When blue litmus is placed in an alkali it stays blue. | When blue litmus is placed in a neutral solution it stays blue. |
References
Edexcel
- Litmus paper, pages 74, 95, 110, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Litmus, page 43, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Litmus, pages 119, 186, 213, 275, 323, GCSE Chemistry, CGP, Edexcel