GCSE Physics Required Practical: Determining Specific Heat Capacity
Revision as of 19:24, 18 March 2019 by NRJC (talk | contribs) (Created page with "==Key Stage 4== ===Meaning=== Determining the specific heat capacity of a metal block. ===Experiment Version 1a=== ====Variables==== : Indepe...")
Contents
Key Stage 4
Meaning
Determining the specific heat capacity of a metal block.
Experiment Version 1a
Variables
- Independent Variable: The temperature of the metal block.
- Dependent Variable: The energy supplied to the metal block by heating.
- Control Variables: The mass of the metal block.
Method
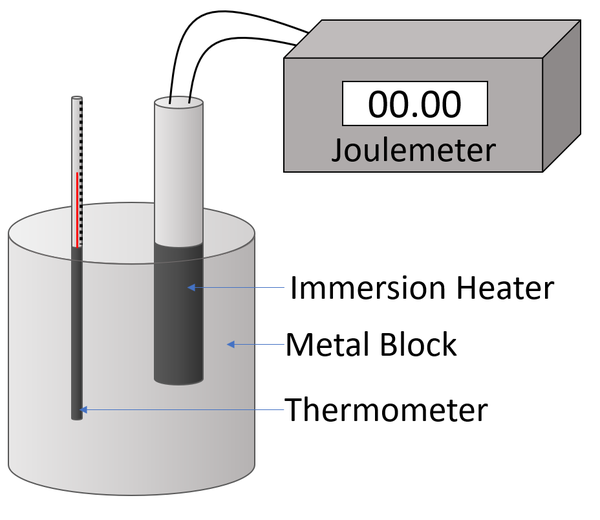
| A diagram of the apparatus used in an experiment to find the specific heat capacity of a metal block. |
- Attach a Joulemeter and power supply to an immersion heater.
- Place the immersion heater and the thermometer in holes in the metal block.
- Place a drop of water in the thermometer hole to ensure thermal contact between the thermometer and the metal block.
- Read and record the initial temperature of the metal block.
- Switch on the power supply.
- Record the reading on the Joulemeter with every 2°C increase in temperature.
Improving Accuracy
- Place the metal block on a heatproof mat to reduce the thermal energy lost to the table surface by conduction.
- Wrap the metal block a thermal insulator to reduce the thermal energy lost to the air.
- Complete the experiment in temperature range close to room temperature to reduce the rate of energy transfer from the metal block to the surroundings.
Improving Precision
- Use a thermometer with a higher resolution.
- Use a data logger rather than a thermometer.