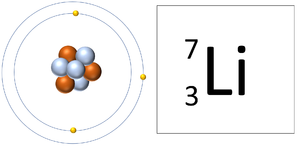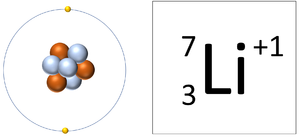Difference between revisions of "Ion"
| Line 10: | Line 10: | ||
{| class="wikitable" | {| class="wikitable" | ||
| + | | style="height:20px; width:300px; text-align:center;" |'''Atom''' | ||
| style="height:20px; width:300px; text-align:center;" |'''Ion''' | | style="height:20px; width:300px; text-align:center;" |'''Ion''' | ||
| − | |||
|- | |- | ||
|[[File:LithiumAtom+Symbol.png|center|300px]] | |[[File:LithiumAtom+Symbol.png|center|300px]] | ||
Revision as of 17:05, 24 November 2018
Key Stage 4
Meaning
An Ion is a particle that has a different number of protons to electrons.
About Ions
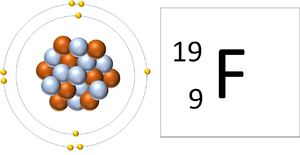
- An atom contains the same number of protons as electrons. When an atom loses or gains electrons it becomes an ion.
- Ions can be positively charged or negatively charged.
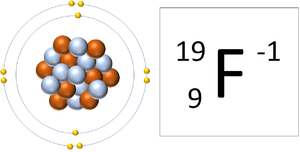
- If an atom gains electrons it becomes a negatively charged ion since there are more electrons than protons and electrons carry a negative charge.
- If an atom loses electrons it becomes a positively charged ion since there are more protons than electrons and protons carry a positive charge.
| Atom | Ion |
| A Lithium atom has 3 protons and 3 electrons so it is neutral. | A Lithium ion has 3 protons and 2 electrons so it has a positive charge. |
| A Fluorine atom has 9 protons and 9 electrons so it is neutral. | A Fluorine atom has 9 protons and 10 electrons so it is negative charge. |