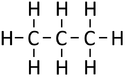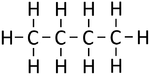Difference between revisions of "Alkane"
(→Complete Combustion) |
|||
| (6 intermediate revisions by one other user not shown) | |||
| Line 65: | Line 65: | ||
: [[Methane]] + [[Oxygen]] → [[Soot]] + [[Water]] | : [[Methane]] + [[Oxygen]] → [[Soot]] + [[Water]] | ||
| − | : < | + | : CH<sub>4</sub> + O<sub>2</sub> → C + 2H<sub>2</sub>O |
: [[Methane]] + [[Oxygen]] → [[Carbon Monoxide]] + [[Water]] | : [[Methane]] + [[Oxygen]] → [[Carbon Monoxide]] + [[Water]] | ||
| − | : < | + | : 2CH<sub>4</sub> + 3O<sub>2</sub> → 2CO + 4H<sub>2</sub>O |
: [[Ethane]] + [[Oxygen]] → [[Soot]] + [[Water]] | : [[Ethane]] + [[Oxygen]] → [[Soot]] + [[Water]] | ||
| − | : < | + | : 2C<sub>2</sub>H<sub>6</sub> + 3O<sub>2</sub> → 4C + 6H<sub>2</sub>O |
: [[Ethane]] + [[Oxygen]] → [[Carbon Monoxide]] + [[Water]] | : [[Ethane]] + [[Oxygen]] → [[Carbon Monoxide]] + [[Water]] | ||
| − | : < | + | : 2C<sub>2</sub>H<sub>6</sub> + 5O<sub>2</sub> → 4CO + 6H<sub>2</sub>O |
: [[Ethane]] + [[Oxygen]] → [[Soot]] + [[Carbon Monoxide]] + [[Water]] | : [[Ethane]] + [[Oxygen]] → [[Soot]] + [[Carbon Monoxide]] + [[Water]] | ||
| − | : < | + | : 2C<sub>2</sub>H<sub>6</sub> + 4O<sub>2</sub> → 2C + 2CO + 6H<sub>2</sub>O |
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945598/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945598&linkCode=as2&tag=nrjc-21&linkId=ad276ad49df77ab4b40ab4fd0fe09666 ''Alkanes, page 146, 148, 149, GCSE Combined Science; The Revision Guide, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Alkanes, pages 124-126, 130, GCSE Combined Science Trilogy; Chemistry, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851362/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851362&linkCode=as2&tag=nrjc-21&linkId=7d78d70a2044ee9982dae010c94af92a ''Alkanes, pages 143-4, GCSE Combined Science Trilogy 2, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Alkanes, pages 149, 160, GCSE Chemistry; Third Edition, Oxford University Press, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Alkanes, pages 172-3, GCSE Chemistry, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Alkanes, pages 188-192, GCSE Combined Science Trilogy; Chemistry, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Alkanes, pages 220 -224, GCSE Chemistry, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Alkanes, pages 228-9, 233, 238-9, 236-41, GCSE Chemistry; Student Book, Collins, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d37 ''Alkanes, pages 75-77, GCSE Chemistry; The Revision Guide, CGP, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851362/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851362&linkCode=as2&tag=nrjc-21&linkId=7d78d70a2044ee9982dae010c94af92a ''Alkanes; Complete combustion of, pages 145-6, GCSE Combined Science Trilogy 2, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851362/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851362&linkCode=as2&tag=nrjc-21&linkId=7d78d70a2044ee9982dae010c94af92a ''Alkanes; Cracking, pages 147-8, GCSE Combined Science Trilogy 2, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Alkanes; cracking, pages 176-7, GCSE Chemistry, Hodder, AQA''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Alkanes; reaction of, 240-1, GCSE Chemistry; Student Book, Collins, AQA'] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Alkanes; structure and formulae, 238-9, GCSE Chemistry; Student Book, Collins, AQA'] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Alkanes; use as fuels, pages 1744-5, GCSE Chemistry, Hodder, AQA'] | ||
| + | ====Edexcel==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Alkane homologous series, pages 268-269, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Alkanes, page 268, GCSE Combined Science, Pearson Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Alkanes, pages 137, 138, 140, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Alkanes, pages 154, 172, 178, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Alkanes, pages 253, 256, 262, 263, 284-287, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Alkanes, pages 87, 88, 90, 98, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Alkanes; homologous series, pages 154-155, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Alkanes; reactions, pages 174-175, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Alkanes, pages 141, 143, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Alkanes, pages 230-231, 238-239, 240, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Alkanes, pages 88, 92-94, GCSE Chemistry; The Revision Guide, CGP, OCR Gateway ''] | ||
Latest revision as of 10:25, 30 November 2019
Contents
Key Stage 4
Meaning
Alkanes are the simplest form of hydrocarbon compounds with no double bonds and the general formula; CnH2n+2
About Alkanes
- Alkanes are a homologous series of hydrocarbon compounds.
- The functional group of the Alkanes is the single bonds between the Carbon atoms and between the Carbon and Hydrogen atoms.
- Alkanes are long chains of Carbon atoms covalently bonded together with single bonds and Hydrogen atoms taking the remaining bonds.
Examples
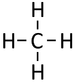
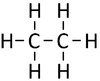
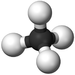
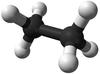
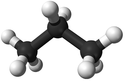
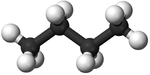
| Methane | Ethane | Propane | Butane | |
| Chemical Formula (CnH2n+2) | CH4 | C2H6 | C3H8 | C4H10 |
| Structural Formula | CH4 | CH3CH3 | CH3CH2CH3 | CH3CH2CH2CH3 |
| Structural Diagram | ||||
| Ball and Stick Model |
Reactions of Alkanes
Combustion
During combustion of alkanes the Carbon and Hydrogen atoms are oxidised to produce Carbon Dioxide and Water.
Complete Combustion
Complete combustion occurs when there is enough Oxygen to completely Oxidise all of the atoms in the alkane. In the complete combustion of alkanes the only products are Carbon Dioxide and Water.
- Methane + Oxygen → Carbon Dioxide + Water
- CH4 + 2O2 → CO2 + 2H2O
- Ethane + Oxygen → Carbon Dioxide + Water
- 2C2H6 + 7O2 → 4CO2 + 6H2O
- Propane + Oxygen → Carbon Dioxide + Water
- C3H8 + 5O2 → 3CO2 + 4H2O
- Butane + Oxygen → Carbon Dioxide + Water
- 2C4H10 + 13O2 → 8CO2 + 10H2O
Incomplete Combustion
Incomplete combustion occurs when there is not enough Oxygen to Oxidise all of the atoms in the alkane. During incomplete combustion of alkanes the products may include Carbon (soot) and Carbon Monoxide.
- Methane + Oxygen → Carbon Monoxide + Water
- 2CH4 + 3O2 → 2CO + 4H2O
- Ethane + Oxygen → Carbon Monoxide + Water
- 2C2H6 + 5O2 → 4CO + 6H2O
- Ethane + Oxygen → Soot + Carbon Monoxide + Water
- 2C2H6 + 4O2 → 2C + 2CO + 6H2O
References
AQA
- Alkanes, page 146, 148, 149, GCSE Combined Science; The Revision Guide, CGP, AQA
- Alkanes, pages 124-126, 130, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Alkanes, pages 143-4, GCSE Combined Science Trilogy 2, Hodder, AQA
- Alkanes, pages 149, 160, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Alkanes, pages 172-3, GCSE Chemistry, Hodder, AQA
- Alkanes, pages 188-192, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Alkanes, pages 220 -224, GCSE Chemistry, CGP, AQA
- Alkanes, pages 228-9, 233, 238-9, 236-41, GCSE Chemistry; Student Book, Collins, AQA
- Alkanes, pages 75-77, GCSE Chemistry; The Revision Guide, CGP, AQA
- Alkanes; Complete combustion of, pages 145-6, GCSE Combined Science Trilogy 2, Hodder, AQA
- Alkanes; Cracking, pages 147-8, GCSE Combined Science Trilogy 2, Hodder, AQA
- Alkanes; cracking, pages 176-7, GCSE Chemistry, Hodder, AQA
- Alkanes; reaction of, 240-1, GCSE Chemistry; Student Book, Collins, AQA'
- Alkanes; structure and formulae, 238-9, GCSE Chemistry; Student Book, Collins, AQA'
- Alkanes; use as fuels, pages 1744-5, GCSE Chemistry, Hodder, AQA'
Edexcel
- Alkane homologous series, pages 268-269, GCSE Combined Science, Pearson Edexcel
- Alkanes, page 268, GCSE Combined Science, Pearson Edexcel
- Alkanes, pages 137, 138, 140, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Alkanes, pages 154, 172, 178, GCSE Chemistry, Pearson, Edexcel
- Alkanes, pages 253, 256, 262, 263, 284-287, GCSE Chemistry, CGP, Edexcel
- Alkanes, pages 87, 88, 90, 98, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Alkanes; homologous series, pages 154-155, GCSE Chemistry, Pearson, Edexcel
- Alkanes; reactions, pages 174-175, GCSE Chemistry, Pearson, Edexcel