Chemical Bond
Contents
Key Stage 4
Meaning
A chemical bond is a force of attraction holding the atoms inside a molecule together.
About Chemical Bonds
There are three types of chemical bond you should know:
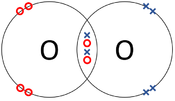
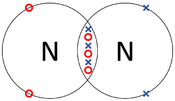
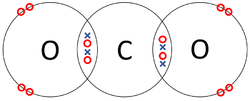
- Covalent Bonds - In which atoms share electrons with one another.
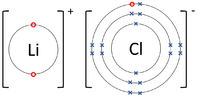
- Ionic Bonds - In which electrons are transferred from one atom to another.
- Metallic Bonds - In which some electrons move freely between atoms creating lattice of positively charged ions surrounded by a sea of delocalised electrons (free electrons).
Examples
| The two Oxygen atoms each share two of their electrons with one another. | The two Nitrogen atoms each share three of their electrons with one another. | Each Oxygen shares two of its electrons with the Carbon atom while the Carbon atom shares two electrons with each Oxygen atom. |
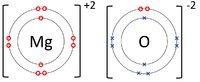
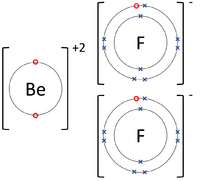
| The Lithium atom donates an electron from its outer shell to the outer shell of the Fluorine atom. | The Magnesium atom donates two electrons from its outer shell to the outer shell of the Oxygen atom. | The Beryllium atom donates two electrons from its outer shell to the outer shells of each Fluorine atom. |
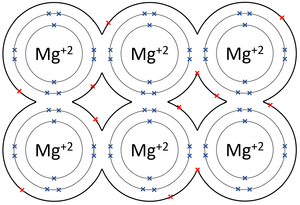
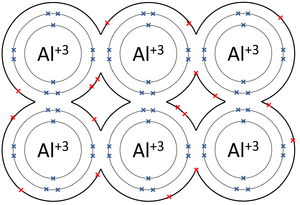
| The outer shells of the Magnesium atoms overlap allowing the two electrons in each outer shell to move freely between atoms. | The outer shells of the Aluminium atoms overlap allowing the three electrons in each outer shell to move freely between atoms. |
Extra Information
References
AQA
- Bonding, page 163, GCSE Combined Science Trilogy 1, Hodder, AQA
- Bonding, pages 47-8, GCSE Chemistry, Hodder, AQA
- Bonding; covalent, pages 154, 156-7, GCSE Combined Science Trilogy 1, Hodder, AQA
- Bonding; covalent, pages 41-2, GCSE Chemistry, Hodder, AQA
- Bonding; ionic, page 149, GCSE Combined Science Trilogy 1, Hodder, AQA
- Bonding; ionic, page 34, GCSE Chemistry, Hodder, AQA
- Bonding; metallic, page 45, GCSE Chemistry, Hodder, AQA
- Bonding; metallic, pages 160-1, GCSE Combined Science Trilogy 1, Hodder, AQA
- Chemical bond, pages 58-9, GCSE Chemistry; Student Book, Collins, AQA
Edexcel
- Bond energies, page 136, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Bond energies, page 85, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bond energies, pages 248-250, GCSE Chemistry, CGP, Edexcel
- Bond energy, page 147, GCSE Chemistry, Pearson, Edexcel
- Bond energy, page 261, GCSE Combined Science, Pearson Edexcel
- Bonding, page 186, GCSE Combined Science, Pearson Edexcel
- Bonding, page 42, GCSE Chemistry, Pearson, Edexcel
- Bonding, pages 21, 23, 25, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding, pages 51-53, 56-61, 65, 66, GCSE Chemistry, CGP, Edexcel
- Bonding, pages 84, 86, 88, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Bonding; covalent, page 23, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding; covalent, pages 184-185, 186, 192, 261, GCSE Combined Science, Pearson Edexcel
- Bonding; covalent, pages 40-41, 42, 48, GCSE Chemistry, Pearson, Edexcel
- Bonding; covalent, pages 56-61, GCSE Chemistry, CGP, Edexcel
- Bonding; giant covalent, page 192, GCSE Combined Science, Pearson Edexcel
- Bonding; giant covalent, page 48, GCSE Chemistry, Pearson, Edexcel
- Bonding; ionic, page 21, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding; ionic, pages 178-179, 192, GCSE Combined Science, Pearson Edexcel
- Bonding; ionic, pages 51-53, GCSE Chemistry, CGP, Edexcel
- Bonding; metallic, page 25, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Bonding; metallic, pages 190, 192, GCSE Combined Science, Pearson Edexcel
- Bonding; metallic, pages 46, 48, GCSE Chemistry, Pearson, Edexcel
- Bonding; metallic, pages 65, 66, GCSE Chemistry, CGP, Edexcel
- Bonding; models, pages 192-193, GCSE Combined Science, Pearson Edexcel
- Bonding; models, pages 48-49, GCSE Chemistry, Pearson, Edexcel
- Bonds; breaking and making, page 146, GCSE Chemistry, Pearson, Edexcel
- Bonds; breaking and making, page 260, GCSE Combined Science, Pearson Edexcel
- Bonds; covalent, page 147, GCSE Chemistry, Pearson, Edexcel
- Bonds; energy calculations, page 147, GCSE Chemistry, Pearson, Edexcel
- Bonds; energy calculations, page 261, GCSE Combined Science, Pearson Edexcel