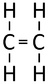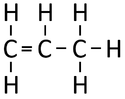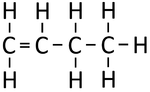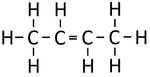Difference between revisions of "Alkene"
(→Examples) |
|||
| Line 41: | Line 41: | ||
|[[File:BallandStickBut-2-ene.png|center|150px]] | |[[File:BallandStickBut-2-ene.png|center|150px]] | ||
|} | |} | ||
| + | |||
| + | ===Reactions of Alkenes=== | ||
| + | ====Combustion==== | ||
| + | During [[combustion]] of [[alkene]]s the [[Carbon]] and [[Hydrogen]] [[atom]]s are [[oxidised]] to produce [[Carbon Dioxide]] and [[Water]]. | ||
| + | =====Complete Combustion===== | ||
| + | [[Complete Combustion|Complete combustion]] occurs when there is enough [[Oxygen]] to completely [[Oxidise]] all of the [[atom]]s in the [[alkene]]. | ||
| + | In the [[Complete Combustion|complete combustion]] of [[alkene]]s the only [[product]]s are [[Carbon Dioxide]] and [[Water]]. | ||
| + | : [[Ethene]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | ||
| + | : <chem>C2H4 + 3O2 -> 2CO2 + 2H2O</chem> | ||
| + | |||
| + | : [[Propene]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | ||
| + | : <chem>2C3H6 + 9O2 -> 6CO2 + 6H2O</chem> | ||
| + | |||
| + | : [[Butene]] + [[Oxygen]] → [[Carbon Dioxide]] + [[Water]] | ||
| + | : <chem>C4H8 + 6O2 -> 4CO2 + 4H2O</chem> | ||
| + | |||
| + | =====Incomplete Combustion===== | ||
| + | [[Incomplete Combustion|Incomplete combustion]] occurs when there is not enough [[Oxygen]] to [[Oxidise]] all of the [[atom]]s in the [[alkene]]. | ||
| + | During [[Incomplete Combustion|incomplete combustion]] of [[alkene]]s the [[product]]s may include [[Carbon]] ([[soot]]) and [[Carbon Monoxide]]. | ||
| + | |||
| + | : [[Ethene]] + [[Oxygen]] → [[Carbon]] + [[Water]] | ||
| + | : <chem>C2H4 + O2 -> 2C + 2H2O</chem> | ||
| + | |||
| + | : [[Ethene]] + [[Oxygen]] → [[Carbon Monoxide]] + [[Water]] | ||
| + | : <chem>C2H4 + 2O2 -> 2CO + 2H2O</chem> | ||
| + | |||
| + | : [[Ethane]] + [[Oxygen]] → [[Carbon]] + [[Carbon Monoxide]] + [[Water]] | ||
| + | : <chem>2C2H4 + 3O2 -> 2C + 2CO + 4H2O</chem> | ||
Revision as of 19:17, 17 January 2019
Contents
Key Stage 4
Meaning
Alkenes are hydrocarbon compounds with one double bond and the general formula; CnH2n
About Alkenes
- Alkenes are a homologous series of hydrocarbon compounds.
- The functional group of the Alkenes is the double bonds between the Carbon atoms.
- Alkenes are long chains of Carbon atoms covalently bonded together with double and single bonds and Hydrogen atoms taking the remaining bonds.
Examples
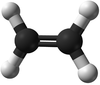
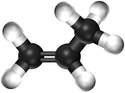
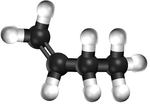
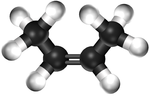
| Ethene | Propene | But-1-ene | But-2-ene | |
| Chemical Formula (CnH2n) | C2H4 | C3H6 | C4H8 | C4H8 |
| Structural Formula | CH2CH2 | CH2CHCH3 | CH2CHCH2CH3 | CH3CHCHCH3 |
| Structural Diagram | ||||
| Ball and Stick Model |
Reactions of Alkenes
Combustion
During combustion of alkenes the Carbon and Hydrogen atoms are oxidised to produce Carbon Dioxide and Water.
Complete Combustion
Complete combustion occurs when there is enough Oxygen to completely Oxidise all of the atoms in the alkene. In the complete combustion of alkenes the only products are Carbon Dioxide and Water.
- Ethene + Oxygen → Carbon Dioxide + Water
- <chem>C2H4 + 3O2 -> 2CO2 + 2H2O</chem>
- Propene + Oxygen → Carbon Dioxide + Water
- <chem>2C3H6 + 9O2 -> 6CO2 + 6H2O</chem>
- Butene + Oxygen → Carbon Dioxide + Water
- <chem>C4H8 + 6O2 -> 4CO2 + 4H2O</chem>
Incomplete Combustion
Incomplete combustion occurs when there is not enough Oxygen to Oxidise all of the atoms in the alkene. During incomplete combustion of alkenes the products may include Carbon (soot) and Carbon Monoxide.
- Ethene + Oxygen → Carbon Monoxide + Water
- <chem>C2H4 + 2O2 -> 2CO + 2H2O</chem>
- Ethane + Oxygen → Carbon + Carbon Monoxide + Water
- <chem>2C2H4 + 3O2 -> 2C + 2CO + 4H2O</chem>