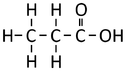Difference between revisions of "Propanoic Acid"
| Line 46: | Line 46: | ||
<math>2C_2H_5COOH + MgCO_3 → (C_2H_5COO)_2Mg + CO_2 + H_2O</math> | <math>2C_2H_5COOH + MgCO_3 → (C_2H_5COO)_2Mg + CO_2 + H_2O</math> | ||
| + | |||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Propanoic acid, page 242, GCSE Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Propanoic acid, pages 244-5, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
Latest revision as of 22:45, 10 November 2019
Contents
Key Stage 3
Meaning
Propanoic Acid is a compound with chemical formula C3H6O2.
About Propanoic Acid
- Propanoic Acid is a solid dissolved in water to form a solution.
Propanoic Acid can be neutralised to produce organic salts.
- Propanoic Acid + Sodium → Sodium Propanoate + Hydrogen
- Propanoic Acid + Potassium Oxide → Potassium Propanoate + Water
- Propanoic Acid + Lithium Hydroxide → Lithium Propanoate + Water
- Propanoic Acid + Magnesium Carbonate → Magnesium Propanoate + Carbon Dioxide + Water
Key Stage 4
Meaning
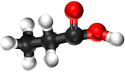
Propanoic Acid is a Carboxylic Acid with chemical formula C3H6O2.
About Propanoic Acid
- Propanoic Acid is an aqueous solution.
| Chemical Formula (CnH2n+2) | Structural Formula | Structural Diagram | Ball and Stick Model |
| C3H6O2 | CH3CH2COOH |
Propanoic Acid can be neutralised to produce organic salts.
\(2C_2H_5COOH + 2Na → 2C_2H_5COONa + H_2\)
\(2C_2H_5COOH + 2K → 2C_2H_5COOK + H_2O\)
\(C_2H_5COOH + LiOH → C_2H_5COOLi + H_2O\)
\(2C_2H_5COOH + MgCO_3 → (C_2H_5COO)_2Mg + CO_2 + H_2O\)