Difference between revisions of "Diamond"
| Line 40: | Line 40: | ||
:[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Diamond, page 24, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Diamond, page 24, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Diamond, page 62, GCSE Chemistry, CGP, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1782948147/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782948147&linkCode=as2&tag=nrjc-21&linkId=f63dcd8345f4e49c717b39a228a36c7c ''Diamond, page 62, GCSE Chemistry, CGP, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Diamond, page 21, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Diamond, page 94, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Diamond, pages 62, 74, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
Latest revision as of 15:13, 4 December 2019
Contents
Key Stage 3
Meaning
Diamonds are large crystal molecules of carbon.
About Diamonds
- Diamonds are very hard.
- Diamonds are transparent.
Key Stage 4
Meaning
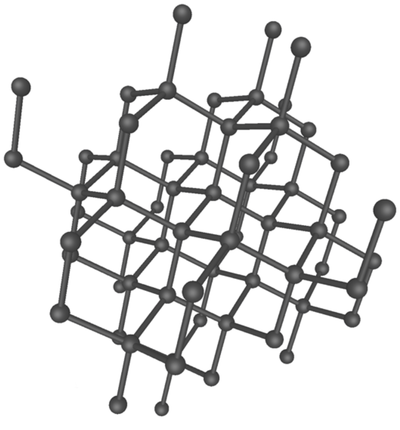
Diamond is an allotrope of carbon in which the atoms are bonded in a tetrahedral arrangement.
| A diagram showing the arrangement of carbon atoms in diamond. |
About Diamonds
- Diamonds are a giant covalent structure.
- Diamonds are extremely hard.
- Diamonds have a high melting point.
- Diamonds are transparent and have a high refractive index.
- Diamond is a poor electrical conductor because there are no free electrons to move around the giant covalent structure.
References
AQA
- Diamond, page 166, GCSE Combined Science Trilogy 1, Hodder, AQA
- Diamond, page 54, GCSE Chemistry, Hodder, AQA
- Diamond, pages 118, 119, GCSE Combined Science; The Revision Guide, CGP, AQA
- Diamond, pages 33, 34, GCSE Chemistry; The Revision Guide, CGP, AQA
- Diamond, pages 48-49, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Diamond, pages 56-7, 64, 76-7, 80-1, 91, GCSE Chemistry; Student Book, Collins, AQA
- Diamond, pages 86, 87, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Diamond, pages 88, 89, GCSE Chemistry, CGP, AQA
Edexcel
- Diamond, page 24, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Diamond, page 62, GCSE Chemistry, CGP, Edexcel