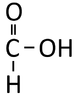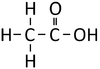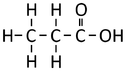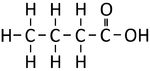Difference between revisions of "Carboxylic Acid"
| Line 13: | Line 13: | ||
| style="height:20px; width:75px; text-align:center;" | | | style="height:20px; width:75px; text-align:center;" | | ||
| style="height:20px; width:150px; text-align:center;" |'''Methanoic Acid''' | | style="height:20px; width:150px; text-align:center;" |'''Methanoic Acid''' | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Carboxylic acids, page 183, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d73 ''Carboxylic acids, page 82, GCSE Chemistry; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Carboxylic acids, pages 161, 163-165, 170-171, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Carboxylic acids, pages 242, 243, GCSE Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Carboxylic acids; production from alcohols, page 182, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Carboxylic acids; properties and reactions of, page 184, GCSE Chemistry, Hodder, AQA ''] | ||
| + | |||
| style="height:20px; width:150px; text-align:center;" |'''Ethanoic Acid''' | | style="height:20px; width:150px; text-align:center;" |'''Ethanoic Acid''' | ||
| style="height:20px; width:150px; text-align:center;" |'''Propanoic Acid''' | | style="height:20px; width:150px; text-align:center;" |'''Propanoic Acid''' | ||
Revision as of 16:57, 2 November 2019
Contents
Key Stage 4
Meaning
Carboxylic Acids are organic compounds with a Carbon atom which has a double bonds to an Oxygen atom and a single bond to an OH group. The general formula is CnH2nO2.
About Carboxylic Acids
- Carboxylic Acids are a homologous series of organic compounds.
- The functional group of the Carboxylic Acids is the double bond between a Carbon atom and an Oxygen atom and the single bond to an OH group.
- Carboxylic Acids are long chains of Carbon atoms covalently bonded together with the the final Carbon atom in the chain as a COOH group.
Examples
Methanoic Acid
ReferencesAQA
|
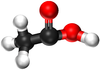
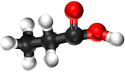
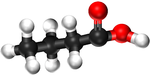
Ethanoic Acid | Propanoic Acid | Butanoic Acid | |
| Chemical Formula | CH2O2 | C2H4O2 | C3H6O2 | C4H8O2 |
| Structural Formula | HCOOH | CH3COOH | CH3CH2COOH | CH3CH2CH2COOH |
| Structural Diagram | ||||
| Ball and Stick Model |
Reactions of Carboxylic Acids
Neutralisation
Carboxylic Acids can be neutralised to produce organic salts.
\(2HCOOH + 2K → 2HCOOK + H_2\)
\(2CH_3COOH + MgO → (CH3COO)_2Mg + H_2O\)
\(C_2H_5COOH + NaOH → C_2H_5COONa + H_2O\)
\(2C_3H_7COOH + MgCO_3 → (C_3H_7COO)_2Mg + H_2O + CO_2\)
Esterification
Carboxylic Acids may react with alcohols to produce compounds known as esters which have the functional group -COO-.
- Methanoic Acid + Ethanol → Ethyl Methanoate + Water
\(HCOOH + C2_H_5OH → HCOOC_2H_5 + H_2O\)
- Ethanoic Acid + Ethanol → Ethyl Ethanoate + Water
\(CH_3COOH + C_2H_5OH → CH_3COOC_2H_5 + H_2O\)
- Propanoic Acid + Propanol → Propyl Propanoate + Water
\(C_3H_5COOH + C_3H_7OH → C_2H_5COOC_3H_7 + H_2O\)
- Butanoic Acid + Propanol → Propyl Butanoate + Water
\(C_3H_5COOH + C_3H_7OH → C_3H_5COOC_3H_7 + H_2O\)