Covalent Bond
Key Stage 4
Meaning
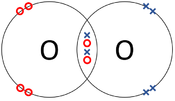
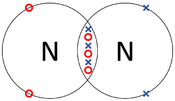
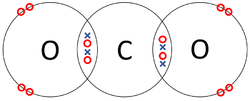
A covalent bond is a type of chemical bond in which atoms share electrons with one another.
About Covalent Bonds
- Atoms are more chemically stable when their outer shell is full of electrons. One way atoms can have a full outer shell is by sharing some electrons with other atoms, this is a covalent bond.
- Covalent bonds happen between non-metal elements.
Examples
| The two Oxygen atoms each share two of their electrons with one another. | The two Nitrogen atoms each share three of their electrons with one another. | Each Oxygen shares two of its electrons with the Carbon atom while the Carbon atom shares two electrons with each Oxygen atom. |