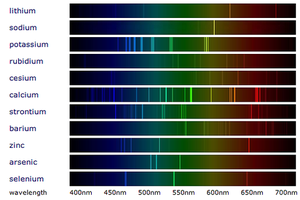
Flame Emission Spectroscopy
Revision as of 12:35, 24 January 2019 by NRJC (talk | contribs) (Created page with "==Key Stage 4== ===Meaning=== right|300px|thumb|The [[Line Spectrum|line spectra from the '''flame emission spectroscopy''' of several [...")
Key Stage 4
Meaning
Flame emission spectroscopy is a technique for identifying metals in a metal compound.
About Flame Emission Spectroscopy
- Flame emission spectroscopy is an advanced version of the Flame Tests and uses a Spectroscope to separate the colours into a spectrum.
- When white light passes through a Spectroscope the colours are split into a spectrum (the rainbow). When metal compounds burn they only produce certain colours so when this light is passed through a spectroscope in flame emission spectroscopy it produces very specific lines called a 'Line Spectrum' instead of the broad spectrum seen from white light.
- Metals in a metal compound can be identified by comparing it to the line spectra of known metals.