Difference between revisions of "Gas"
(→Example Calculations) |
(→Example Calculations) |
||
| Line 89: | Line 89: | ||
|+ Finding the Volume of Gas | |+ Finding the Volume of Gas | ||
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''Calculate the [[Volume (Space)|volume]] of 4g of [[Hydrogen]] [[gas]].''' |
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''Calculate the [[Volume (Space)|volume]] of 8g of [[Oxygen]] [[gas]].''' |
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''Calculate the [[Volume (Space)|volume]] of 20g of [[Methane]] [[gas]].''' |
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |1.'''Find the [[Relative Formula Mass]] of the [[gas]].''' |
[[Chemical Formula]] of [[Hydrogen]] = H<sub>2</sub> | [[Chemical Formula]] of [[Hydrogen]] = H<sub>2</sub> | ||
| Line 101: | Line 101: | ||
[[Relative Formula Mass]] of H<sub>2</sub> = 2g | [[Relative Formula Mass]] of H<sub>2</sub> = 2g | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |1.'''Find the [[Relative Formula Mass]] of the [[gas]].''' |
[[Chemical Formula]] of [[Oxygen]] = O<sub>2</sub> | [[Chemical Formula]] of [[Oxygen]] = O<sub>2</sub> | ||
| Line 108: | Line 108: | ||
[[Relative Formula Mass]] of O<sub>2</sub> = 32g | [[Relative Formula Mass]] of O<sub>2</sub> = 32g | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |1.'''Find the [[Relative Formula Mass]] of the [[gas]].''' |
[[Chemical Formula]] of [[Methane]] = CH<sub>4</sub> | [[Chemical Formula]] of [[Methane]] = CH<sub>4</sub> | ||
| Line 119: | Line 119: | ||
[[Relative Formula Mass]] of CH<sub>4</sub> = 16g | [[Relative Formula Mass]] of CH<sub>4</sub> = 16g | ||
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''2. Calculate the number of [[mole]]s of [[gas]].''' |
1 [[mole]] = 2g | 1 [[mole]] = 2g | ||
| Line 128: | Line 128: | ||
No. [[Mole]]s = 2mol | No. [[Mole]]s = 2mol | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''2. Calculate the number of [[mole]]s of [[gas]].''' |
1 [[mole]] = 32g | 1 [[mole]] = 32g | ||
| Line 137: | Line 137: | ||
No. [[Mole]]s = 0.25 mol | No. [[Mole]]s = 0.25 mol | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''2. Calculate the number of [[mole]]s of [[gas]].''' |
1 [[mole]] = 16g | 1 [[mole]] = 16g | ||
| Line 147: | Line 147: | ||
No. [[Mole]]s = 1.25 mol | No. [[Mole]]s = 1.25 mol | ||
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''3. Find the [[Volume (Space)|volume]] in dm<sup>3</sup>.''' |
[[Volume (Space)|Volume]] = 24 x (number of moles) | [[Volume (Space)|Volume]] = 24 x (number of moles) | ||
| Line 154: | Line 154: | ||
[[Volume (Space)|Volume]] = 48dm<sup>3</sup> | [[Volume (Space)|Volume]] = 48dm<sup>3</sup> | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''3. Find the [[Volume (Space)|volume]] in dm<sup>3</sup>.''' |
[[Volume (Space)|Volume]] = 24 x (number of moles) | [[Volume (Space)|Volume]] = 24 x (number of moles) | ||
| Line 161: | Line 161: | ||
[[Volume (Space)|Volume]] = 6dm<sup>3</sup> | [[Volume (Space)|Volume]] = 6dm<sup>3</sup> | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''3. Find the [[Volume (Space)|volume]] in dm<sup>3</sup>.''' |
[[Volume (Space)|Volume]] = 24 x (number of moles) | [[Volume (Space)|Volume]] = 24 x (number of moles) | ||
| Line 173: | Line 173: | ||
|+ Finding the Mass of Gas from the Volume | |+ Finding the Mass of Gas from the Volume | ||
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''Calculate the [[mass]] of 12dm<sup>3</sup> of [[Hydrogen]] [[gas]].''' |
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''Calculate the [[mass]] of 36dm<sup>3<sup> of [[Nitrogen]] [[gas]].''' |
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''Calculate the [[mass]] of 18dm<sup>3</sup> of [[Ethane]] [[gas]].''' |
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |1.'''Find the [[Relative Formula Mass]] of the [[gas]].''' |
[[Chemical Formula]] of [[Hydrogen]] = H<sub>2</sub> | [[Chemical Formula]] of [[Hydrogen]] = H<sub>2</sub> | ||
| Line 185: | Line 185: | ||
[[Relative Formula Mass]] of H<sub>2</sub> = 2g | [[Relative Formula Mass]] of H<sub>2</sub> = 2g | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |1.'''Find the [[Relative Formula Mass]] of the [[gas]].''' |
[[Chemical Formula]] of [[Nitrogen]] = N<sub>2</sub> | [[Chemical Formula]] of [[Nitrogen]] = N<sub>2</sub> | ||
| Line 192: | Line 192: | ||
[[Relative Formula Mass]] of N<sub>2</sub> = 28g | [[Relative Formula Mass]] of N<sub>2</sub> = 28g | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |1.'''Find the [[Relative Formula Mass]] of the [[gas]].''' |
[[Chemical Formula]] of [[Ethane]] = C<sub>2</sub>H<sub>6</sub> | [[Chemical Formula]] of [[Ethane]] = C<sub>2</sub>H<sub>6</sub> | ||
| Line 202: | Line 202: | ||
[[Relative Formula Mass]] of C<sub>2</sub>H<sub>6</sub> = 30g | [[Relative Formula Mass]] of C<sub>2</sub>H<sub>6</sub> = 30g | ||
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''2. Calculate the number of [[mole]]s of [[gas]].''' |
[[Volume (Space)|Volume]] = 24 x (number of moles) | [[Volume (Space)|Volume]] = 24 x (number of moles) | ||
| Line 211: | Line 211: | ||
Number of moles = 0.5 | Number of moles = 0.5 | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''2. Calculate the number of [[mole]]s of [[gas]].''' |
[[Volume (Space)|Volume]] = 24 x (number of moles) | [[Volume (Space)|Volume]] = 24 x (number of moles) | ||
| Line 220: | Line 220: | ||
Number of moles = 1.5 | Number of moles = 1.5 | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''2. Calculate the number of [[mole]]s of [[gas]].''' |
[[Volume (Space)|Volume]] = 24 x (number of moles) | [[Volume (Space)|Volume]] = 24 x (number of moles) | ||
| Line 230: | Line 230: | ||
Number of moles = 0.75 | Number of moles = 0.75 | ||
|- | |- | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''3. Find the [[mass]] of [[gas]] from the number of [[mole]]s.''' |
No. [[Mole]]s = <math>\frac{Mass}{M_r}</math> | No. [[Mole]]s = <math>\frac{Mass}{M_r}</math> | ||
| Line 240: | Line 240: | ||
Mass = 1g | Mass = 1g | ||
[[Volume (Space)|Volume]] = 48dm<sup>3</sup> | [[Volume (Space)|Volume]] = 48dm<sup>3</sup> | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''3. Find the [[mass]] of [[gas]] from the number of [[mole]]s.''' |
No. [[Mole]]s = <math>\frac{Mass}{M_r}</math> | No. [[Mole]]s = <math>\frac{Mass}{M_r}</math> | ||
| Line 249: | Line 249: | ||
Mass = 42g | Mass = 42g | ||
| − | | style="height:20px; width: | + | | style="height:20px; width:250px; text-align:center;" |'''3. Find the [[mass]] of [[gas]] from the number of [[mole]]s.''' |
No. [[Mole]]s = <math>\frac{Mass}{M_r}</math> | No. [[Mole]]s = <math>\frac{Mass}{M_r}</math> | ||
Revision as of 14:23, 23 January 2019
Contents
Key Stage 2
Meaning
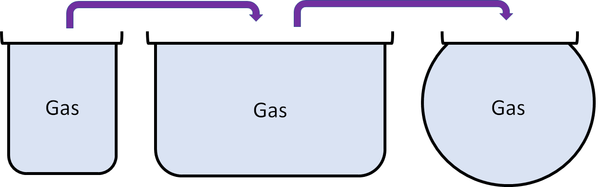
Gas is a state of matter that can change size and shape to fit any container.
About Gases
- Most gases are invisible but we can feel them.
- When the air moves we call it the wind.
|
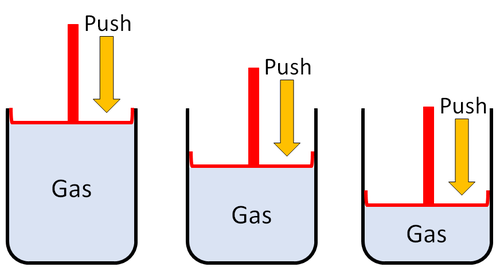
| Gases can be squashed into a smaller size. |
Examples of gas materials:
- Air (A mixture of gases, mostly nitrogen and oxygen)
- Steam
Key Stage 3
Meaning
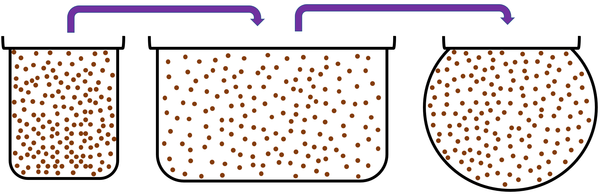
Gas is a State of Matter in which the particles are separated by large distances and can move freely.
About Gases
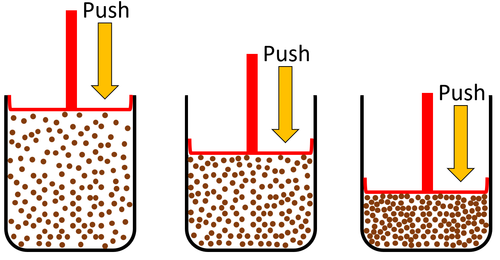
| Gas can be squashed into a smaller size because the particles are spread apart. |
Key Stage 4
Meaning
Gas is a State of Matter in which the particles are separated by large distances and can move freely.
About Gases
- When a substance is in its gaseous state it is always less dense than in its liquid or solid state due to the particles in a gas being spread far apart from each other.
- A substance which is gaseous at room temperature has a smaller force of attraction between particles than a substance which is liquid or solid at room temperature.
| Particle Diagram | Particle Arrangement | Property |
| Particles are free to move in all directions. | Gases fit the size of their container. | |
| Gases fit the shape of their container. | ||
| Convection happens most easily in gases. | ||
| Particles are spread apart. | Gases can be compressed into a smaller volume. | |
| Sound passes through gases slower than liquids and solids. | ||
| Thermal Conduction is very poor in a gases. |
Gas Volume and Mass
- At Standard Temperature and Pressure (20°C and 101,000Pa) the volume of one mole of gas is 24 dm3.
- The volume of gas required or produced in a chemical reaction can be calculated from the mass of the gas and its Relative Formula Mass.
Example Calculations
| Calculate the volume of 4g of Hydrogen gas. | Calculate the volume of 8g of Oxygen gas. | Calculate the volume of 20g of Methane gas. |
| 1.Find the Relative Formula Mass of the gas.
Chemical Formula of Hydrogen = H2 Relative Atomic Mass of Hydrogen = 1g Relative Formula Mass of H2 = 2g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Oxygen = O2 Relative Atomic Mass of Oxygen = 16g Relative Formula Mass of O2 = 32g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Methane = CH4 Relative Atomic Mass of Hydrogen = 1g
Relative Formula Mass of CH4 = 16g |
| 2. Calculate the number of moles of gas.
1 mole = 2g No. Moles = \(\frac{Mass}{M_r}\) No. Moles = \(\frac{4}{2}\) No. Moles = 2mol |
2. Calculate the number of moles of gas.
1 mole = 32g No. Moles = \(\frac{Mass}{M_r}\) No. Moles = \(\frac{8}{32}\) No. Moles = 0.25 mol |
2. Calculate the number of moles of gas.
1 mole = 16g No. Moles = \(\frac{Mass}{M_r}\) No. Moles = \(\frac{20}{16}\) No. Moles = 1.25 mol |
| 3. Find the volume in dm3.
Volume = 24 x (number of moles) Volume = 24 x (2) Volume = 48dm3 |
3. Find the volume in dm3.
Volume = 24 x (number of moles) Volume = 24 x (0.25) Volume = 6dm3 |
3. Find the volume in dm3.
Volume = 24 x (number of moles) Volume = 24 x (1.25) Volume = 30dm3 |
| Calculate the mass of 12dm3 of Hydrogen gas. | Calculate the mass of 36dm3 of Nitrogen gas. | Calculate the mass of 18dm3 of Ethane gas. |
| 1.Find the Relative Formula Mass of the gas.
Chemical Formula of Hydrogen = H2 Relative Atomic Mass of Hydrogen = 1g Relative Formula Mass of H2 = 2g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Nitrogen = N2 Relative Atomic Mass of Nitrogen = 14g Relative Formula Mass of N2 = 28g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Ethane = C2H6 Relative Atomic Mass of Hydrogen = 1g Relative Atomic Mass of Carbon = 12g Relative Formula Mass of C2H6 = 30g |
| 2. Calculate the number of moles of gas.
Volume = 24 x (number of moles) 12 = 24 x (number of moles) Number of moles = 12/24 Number of moles = 0.5 |
2. Calculate the number of moles of gas.
Volume = 24 x (number of moles) 36 = 24 x (number of moles) Number of moles = 36/24 Number of moles = 1.5 |
2. Calculate the number of moles of gas.
Volume = 24 x (number of moles) 12 = 24 x (number of moles) Number of moles = 18/24 Number of moles = 0.75 |
| 3. Find the mass of gas from the number of moles.
No. Moles = \(\frac{Mass}{M_r}\) 0.5 = \(\frac{Mass}{2}\) Mass = \(0.5\times2\) Mass = 1g Volume = 48dm3 |
3. Find the mass of gas from the number of moles.
No. Moles = \(\frac{Mass}{M_r}\) 1.5 = \(\frac{Mass}{28}\) Mass = \(1.5\times28\) Mass = 42g |
3. Find the mass of gas from the number of moles.
No. Moles = \(\frac{Mass}{M_r}\) 0.75 = \(\frac{Mass}{30}\) Mass = \(0.75\times30\) Mass = 22.5g |