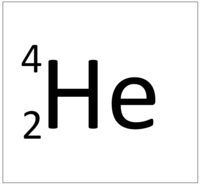Difference between revisions of "Helium"
(→Key Stage 4) |
|||
| Line 21: | Line 21: | ||
: [[Helium]] [[gas]] is less [[density|dense]] than [[air]]. | : [[Helium]] [[gas]] is less [[density|dense]] than [[air]]. | ||
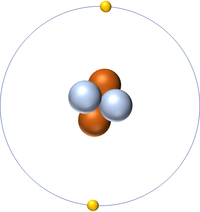
: An [[atom]] of [[Helium]] has two [[electron]]s in its [[Outer Shell|outer shell]]. | : An [[atom]] of [[Helium]] has two [[electron]]s in its [[Outer Shell|outer shell]]. | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |[[File:Helium.png|center|200px]] | ||
| + | |[[File:HeliumSymbol.png|center|200px]] | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" colspan = "2"|[[Helium]] always has 2 [[proton]]s. The most [[Stable Isotope|stable]] [[isotope]] has 2 [[neutron]]s. | ||
| + | |} | ||
Revision as of 12:22, 31 March 2019
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
Helium is a Noble Gas with 2 protons in the nucleus.
About Helium
- Helium has the chemical formula He.
- Helium is inert.
- Helium is a gas at room temperature.
- Helium gas is less dense than air.
- An atom of Helium has two electrons in its outer shell.
Key Stage 4
Meaning
Helium is a Noble Gas with 2 protons in the nucleus.
About Helium
- Helium has the chemical formula He.
- Helium is inert.
- Helium is a gas at standard temperature and pressure.
- Helium gas is less dense than air.
- An atom of Helium has two electrons in its outer shell.
| Helium always has 2 protons. The most stable isotope has 2 neutrons. | |