Difference between revisions of "Solution"
| Line 63: | Line 63: | ||
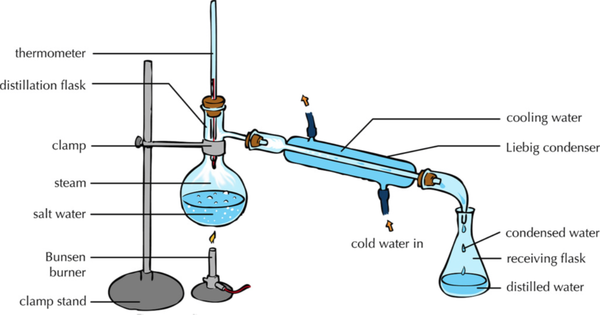
| style="height:20px; width:600px; text-align:center;" |This [[diagram]] shows how the [[solute]] and [[solvent]] can be [[Separating Mixtures|separated]] by heating the [[solution]] to [[evaporate]] the [[solvent]] leaving the [[solute]] behind and then [[condensing]] the [[evaporate]]d [[solvent]] by cooling it down and collecting it in a [[Conical Flask|conical flask]]. | | style="height:20px; width:600px; text-align:center;" |This [[diagram]] shows how the [[solute]] and [[solvent]] can be [[Separating Mixtures|separated]] by heating the [[solution]] to [[evaporate]] the [[solvent]] leaving the [[solute]] behind and then [[condensing]] the [[evaporate]]d [[solvent]] by cooling it down and collecting it in a [[Conical Flask|conical flask]]. | ||
|} | |} | ||
| + | |||
| + | ==Key Stage 4== | ||
| + | ===Meaning=== | ||
| + | : A '''solution''' is a [[mixture]] where a [[substance]] (usually a [[solid]]) is [[dissolve]]d in a [[liquid]]. | ||
| + | |||
| + | ===About Solutions=== | ||
| + | : [[Solution]]s are usually made of a [[liquid]] that is known as a [[solvent]] and a [[solid]] which is known as a [[solute]]. | ||
| + | : When a [[solute]] [[dissolve]]s in a [[solvent]] it becomes a [[solution]]. | ||
| + | : Some [[solution]]s are a [[mixture]] of two [[solvent]]s, for example [[water]] and [[ethanol]]. | ||
| + | : Some [[gas]]es may also [[dissolve]] in a [[liquid]] to make a [[solution]]. For example; [[Carbon Dioxide]] may [[dissolve]] in [[water]]. This makes the [[water]] [[acid]]ic and is what leads to [[Ocean]] [[acid]]ification if there is too much [[Carbon Dioxide]] in the [[Earth's Atmosphere|atmosphere]]. If you have drunk a 'carbonated' beverage (like cola or lemonade) you will have seen the [[Carbon Dioxide]] coming out of [[solution]] and creating bubbles. This is what makes the drink 'fizzy'. | ||
| + | |||
===References=== | ===References=== | ||
Revision as of 15:12, 7 January 2021
Contents
Key Stage 2
Meaning
A solution is a mixture where a substance is dissolved in a liquid.
About Solutions
- For most solutions the liquid is water.
- Some solids dissolve in water to make a solution.
- Once a substance is dissolved in water it is often not possible to see it anymore.
- If a substance has a colour before it is dissolved the solution be will that colour.
Examples
| Salt | Sugar |
| Salt dissolves in water to make a solution called Brine. | Sugar dissolves in water to make a solution. |
Key Stage 3
Meaning
About Solutions
- Solutions are usually made of a liquid that is known as a solvent and a solid which is known as a solute.
- When a solute dissolves in a solvent it becomes a solution.
- Some solutions are a mixture of two solvents, for example water and ethanol.
Examples
| Brown sugar is the solute. | Water is the solvent. | The solute dissolves in the solvent to make a solution. |
| Copper Sulphate is the solute. | Water is the solvent. | The solute dissolves in the solvent to make a solution. |
Separating Solutions
- The solute and solvent can be separated by distillation.
| This diagram shows how the solute and solvent can be separated by heating the solution to evaporate the solvent leaving the solute behind and then condensing the evaporated solvent by cooling it down and collecting it in a conical flask. |
Key Stage 4
Meaning
About Solutions
- Solutions are usually made of a liquid that is known as a solvent and a solid which is known as a solute.
- When a solute dissolves in a solvent it becomes a solution.
- Some solutions are a mixture of two solvents, for example water and ethanol.
- Some gases may also dissolve in a liquid to make a solution. For example; Carbon Dioxide may dissolve in water. This makes the water acidic and is what leads to Ocean acidification if there is too much Carbon Dioxide in the atmosphere. If you have drunk a 'carbonated' beverage (like cola or lemonade) you will have seen the Carbon Dioxide coming out of solution and creating bubbles. This is what makes the drink 'fizzy'.
References
AQA
- Solution, pages 112-3, GCSE Chemistry; Student Book, Collins, AQA
- Solutions, concentrations of, page 193, GCSE Combined Science Trilogy 1, Hodder, AQA
- Solutions, concentrations of, pages 85-7, GCSE Chemistry, Hodder, AQA
- Solutions; concentrations, pages 72-73, I GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Solutions; electrolysis, page 103, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Solutions; ionic compounds, pages 42-43, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Solutions; mass of solutes, page 73, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Solutions; titrations, pages 74-77, GCSE Chemistry; Third Edition, Oxford University Press, AQA
Edexcel
- Solutions, pages 6, 74, GCSE Chemistry, Pearson, Edexcel
- Solutions; concentrated, page 54, GCSE Chemistry, Pearson, Edexcel
- Solutions; dilute, page 54, GCSE Chemistry, Pearson, Edexcel
- Solutions; saturated, page 6, GCSE Chemistry, Pearson, Edexcel