Difference between revisions of "Electronic Structure"
(Created page with "==Key Stage 4== ===Meaning=== The '''electronic configuration''' of an atom is the way the electrons are arranged in orbitals around the Atomic...") |
|||
| Line 15: | Line 15: | ||
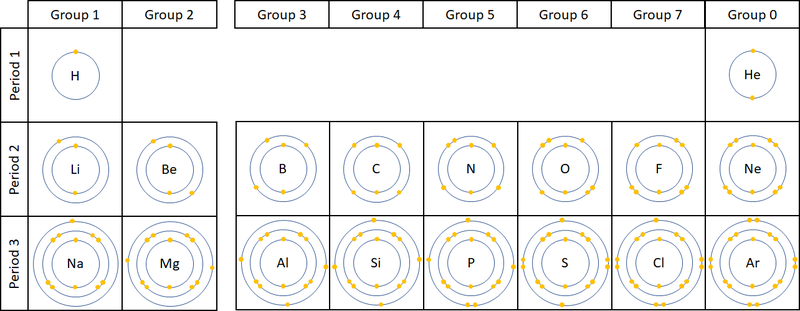
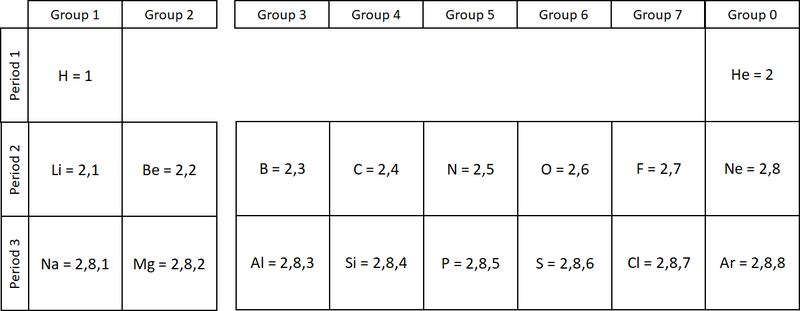
| style="height:20px; width:200px; text-align:center;" |The two [[diagram]]s show how [[electron]]s are '''configured''' in the first 20 [[element]]s on the [[Periodic Table]]. | | style="height:20px; width:200px; text-align:center;" |The two [[diagram]]s show how [[electron]]s are '''configured''' in the first 20 [[element]]s on the [[Periodic Table]]. | ||
|} | |} | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Electronic structure, page 121, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Electronic structure, pages 28-31, 164, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Electronic structure; and the periodic table, pages 127-8, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0008158762/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0008158762&linkCode=as2&tag=nrjc-21&linkId=a0fffa35b3ea49a63404f6704e0df7cc ''Electronic structure; stable, pages 38, 43, 59, 60-1, 164-5, GCSE Chemistry; Student Book, Collins, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Electronic structures, pages 12-14, 18-19, 24-25, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d111 ''Electronic structures, pages 20, 23, 28, GCSE Chemistry; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Electronic structures, pages 44, 45, 71, GCSE Combined Science Trilogy; Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945962/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945962&linkCode=as2&tag=nrjc-21&linkId=476bb5c8d1dfb5c08ac81b6d4d1c98d8 ''Electronic structures, pages 44, 45, 73, GCSE Chemistry, CGP, AQA ''] | ||
Revision as of 11:00, 4 November 2019
Key Stage 4
Meaning
The electronic configuration of an atom is the way the electrons are arranged in orbitals around the nucleus.
About The Electronic Configuration
- The electronic configuration of an atom is written by stating the number of electrons in each electron shell.
- As an example Argon has 2 electrons in the first shell, 8 in the second and 8 in the last, so the electronic configuration is 2,8,8.
| The two diagrams show how electrons are configured in the first 20 elements on the Periodic Table. |
References
AQA
- Electronic structure, page 121, GCSE Combined Science Trilogy 1, Hodder, AQA
- Electronic structure, pages 28-31, 164, GCSE Chemistry; Student Book, Collins, AQA
- Electronic structure; and the periodic table, pages 127-8, GCSE Combined Science Trilogy 1, Hodder, AQA
- Electronic structure; stable, pages 38, 43, 59, 60-1, 164-5, GCSE Chemistry; Student Book, Collins, AQA
- Electronic structures, pages 12-14, 18-19, 24-25, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Electronic structures, pages 20, 23, 28, GCSE Chemistry; The Revision Guide, CGP, AQA
- Electronic structures, pages 44, 45, 71, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Electronic structures, pages 44, 45, 73, GCSE Chemistry, CGP, AQA