Periodic Table
Key Stage 3
Meaning
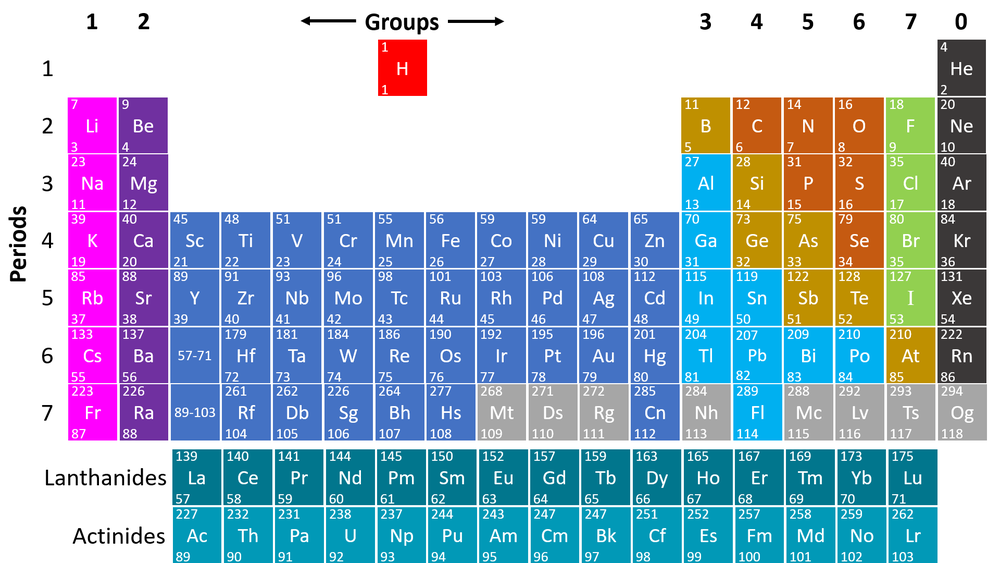
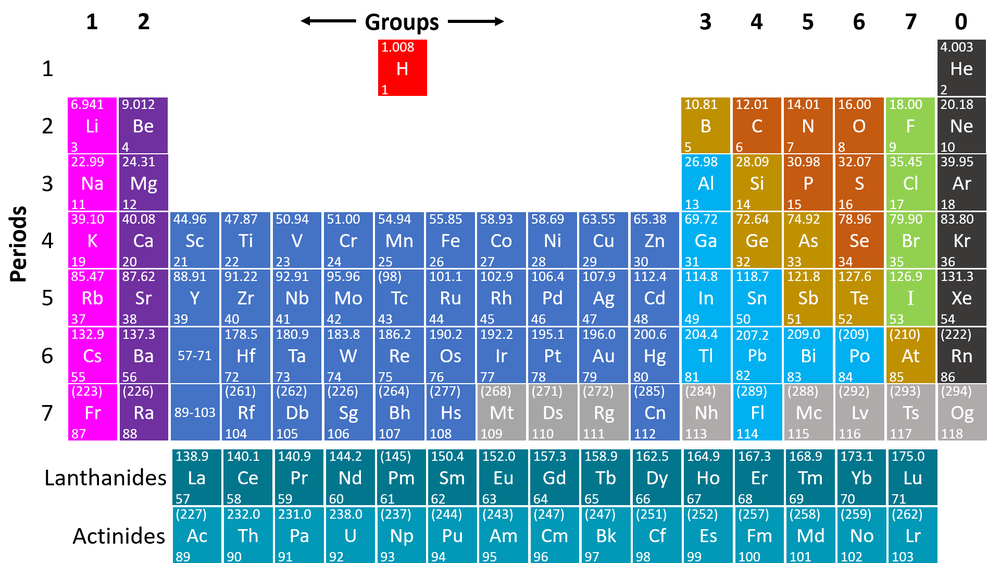
The Periodic Table is a chart listing all the known elements arranged in order of Atomic Number and in columns of elements with similar properties.
About The Periodic Table
- The modern Periodic Table is based on an earlier Periodic Table made by a scientist called Mendeleev. Others had tried to arrange all the elements before, but Mendeleev was the first to correctly arrange it by both Relative Atomic Mass and chemical properties with gaps where elements were later discovered.
- The modern Periodic Table is only slightly different and is arranged by Atomic Number and chemical properties.
- The columns of the Periodic Table are called groups.
- The rows of the Periodic Table are called periods.
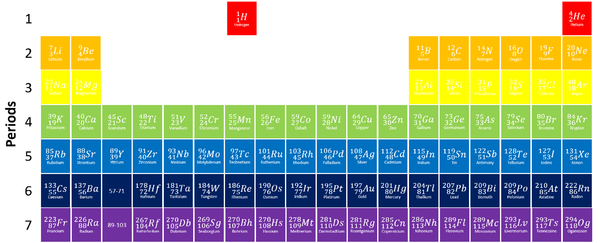
Groups
- The elements are arranged groups of similar chemical properties.
- Elements have similar chemical properties when they have the same number of electrons in the Outer Shell.
Trends within groups
The chemical properties of elements within a group are similar. However, the reactivity within a group changes as you move up or down the periods.
- Group 1: The Alkali Metals all react strongly with water. The reactivity increases as you go down the group.
- Group 2: The Alkali Earth Metals all react strongly with steam and acids. The reactivity increases as you go down the group.
- Group 7: The Halogens all act as bleaching agents and kill bacteria. The reactivity decreases as you go down the group.
- Group 0: The Noble Gases are all inert (unreactive).
The physical properties of elements within a group are similar. However, the property changes gradually as you move down the group.
Periods
- The periods are arranged by the number of Electron Shells.
Trends within Periods
- The chemical and physical properties of elements change as you move along a period.
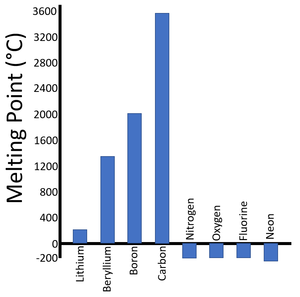
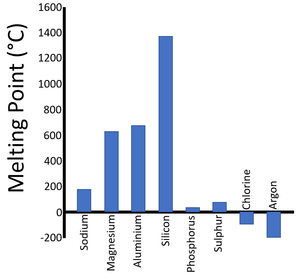
| Period 2 Melting Points | Period 3 Melting Points |
| There is a trend in the Melting Points as you move along the period. | A similar trend can be seen in the next period. |
Periodic Table Song
Linked Periodic Table
Key Stage 4
Meaning
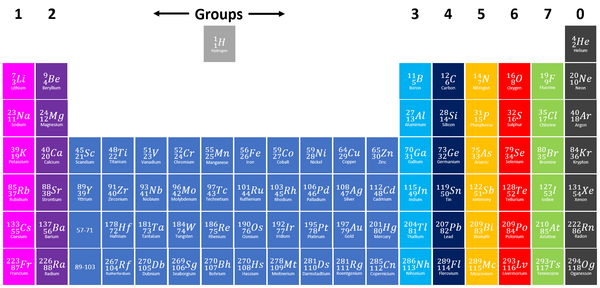
The Periodic Table is a chart listing all the known elements arranged in order of Atomic Number and in columns of elements with similar properties.
About The Periodic Table
- The modern Periodic Table has the elements arranged by Atomic Number and chemical properties.
- Elements on the Periodic Table are written with their Chemical Symbols and two numbers representing their Relative Atomic Mass and Atomic Number.
- The Relative Atomic Masses on the Periodic Table are an average atomic mass of different isotopes of the same element.
- The position of an element in the Periodic Table is a result of the Electronic Structure of the element, which itself is due to the Atomic Number of the element.
- The elements in each column of the Periodic Table all have the same number of electrons in the Outer Shell (except Helium in the Noble Gases) which causes them all to have the same chemical properties. These columns are known as groups.
- The elements in each row of the Periodic Table all have the same number of Electron Shells. These rows are called periods.
History of the Periodic Table
- Scientists always try to look for patterns to help arrange complicated problems. Many different elements had been discovered so scientists wanted to arrange them based on patterns they noticed. Some scientists arranged them by patterns in chemical properties while others tried to arrange them by patterns in physical properties like melting point or relative atomic mass.
- In 1808 a scientist called John Dalton, who created the Dalton Model of the atom, arranged elements by their relative atomic mass.
- In 1864 a scientist called John Newlands arranged elements by relative atomic mass but because he noticed that ever 8th element had similar chemical properties he arranged them into what he called 'octaves'. However, he had not taken into account the possibility that some elements had not been discovered, so he placed some elements into the wrong groups just to fit the pattern he thought he had seen.
- In 1869 Mendeleev was the first to place gaps where the elements did not fit the pattern but still arranged them by both their chemical properties and relative atomic mass. This enabled Mendeleev to predict the existence of elements that had not yet been discovered.
- Mendeleev's Periodic Table was proven (nearly) correct when new elements were discovered that fit in the the gaps he had left. This ability of his table to predict the properties of new elements was what convinced other scientists his Periodic Table was (nearly) correct.
- There were some problems with Mendeleev's Periodic Table that kept scientists skeptical of it. One such problem was that Argon has a greater relative atomic mass than Potassium which would put Argon in the Alkali Metals.
- Once the structure of the atom was investigated by physicists in the early 1900s it was discovered how to correct Mendeleev's Periodic Table by ordering the elements by atomic number instead of relative atomic mass. This produced the modern Periodic Table.
Extra Information
Linked Periodic Table
References
AQA
- Periodic table, page 21, 22, GCSE Chemistry; The Revision Guide, CGP, AQA
- Periodic table, page 9, GCSE Chemistry, Hodder, AQA
- Periodic table, pages 106, 107, GCSE Combined Science; The Revision Guide, CGP, AQA
- Periodic table, pages 109, 117, GCSE Physics; Student Book, Collins, AQA
- Periodic table, pages 12-3, 28, 30-33, 36-7, 44-9, GCSE Chemistry; Student Book, Collins, AQA
- Periodic table, pages 125, 357, GCSE Combined Science Trilogy 1, Hodder, AQA
- Periodic table, pages 287, GCSE Combined Science Trilogy 2, Hodder, AQA
- Periodic table, pages 4-5, 19,22-35, 40-41, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Periodic table, pages 50-53, GCSE Chemistry, CGP, AQA
- Periodic table, pages 50-53, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Periodic table; and electron structure, page 12, GCSE Chemistry, Hodder, AQA
- Periodic table; and electronic structure, pages 127-8, GCSE Combined Science Trilogy 1, Hodder, AQA
- Periodic table; Group 0 (noble gases), page 13, GCSE Chemistry, Hodder, AQA
- Periodic table; Group 0 (noble gases), pages 128-9, GCSE Combined Science Trilogy 1, Hodder, AQA
- Periodic table; Group 1 (alkali metals), page 129-131, 162, GCSE Combined Science Trilogy 1, Hodder, AQA
- Periodic table; Group 1 (alkali metals), pages 14-15, GCSE Chemistry, Hodder, AQA
- Periodic table; Group 7 (halogens), pages 131-4, GCSE Combined Science Trilogy 1, Hodder, AQA
- Periodic table; Group 7 (halogens), pages 16-18, GCSE Chemistry, Hodder, AQA
- Periodic table; group trends, pages 40-7, GCSE Chemistry; Student Book, Collins, AQA
- Periodic table; history of, pages 134-6, GCSE Combined Science Trilogy 1, Hodder, AQA
- Periodic table; history of, pages 19-21, 30-1, GCSE Chemistry, Hodder, AQA
- Periodic table; period, pages 30, 46, GCSE Chemistry; Student Book, Collins, AQA
- Periodic table; table, group, pages 30, 40-5, GCSE Chemistry; Student Book, Collins, AQA
- Periodic table; transition metals, pages 18-19, GCSE Chemistry, Hodder, AQA
Edexcel
- Periodic table, page 18, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Periodic table, page 20, GCSE Chemistry, Pearson, Edexcel
- Periodic table, page 81, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Periodic table, pages 39-41, GCSE Chemistry, CGP, Edexcel
- Periodic table; atomic number, pages 28-29, GCSE Chemistry, Pearson, Edexcel
- Periodic table; electronic configurations, pages 30-31, GCSE Chemistry, Pearson, Edexcel
- Periodic table; elements, pages 26-27, GCSE Chemistry, Pearson, Edexcel
- Periodic table; group 0 elements, pages 134-135, GCSE Chemistry, Pearson, Edexcel
- Periodic table; group 1 elements, pages 128-129, GCSE Chemistry, Pearson, Edexcel
- Periodic table; group 7 elements, pages 130-131, 132-133, GCSE Chemistry, Pearson, Edexcel
- Periodic table; valency, page 41, GCSE Chemistry, Pearson, Edexcel
OCR
- Periodic table, page 16, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR
- Periodic table, page 87, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR
- Periodic Table, pages 36, 52-55, 68-71, 86-88, 132-143, Gateway GCSE Chemistry, Oxford, OCR
Beyond the Curriculum
Isotopes and Atomic Mass
- While the periodic table provides a snapshot of elements based on their atomic number, it's essential to understand the concept of isotopes. Isotopes are variants of an element with the same number of protons but different numbers of neutrons. This variation in neutron count results in different atomic masses. Therefore, the atomic mass on the periodic table represents an average of these isotopes' masses. Exploring isotopes can deepen your understanding of an element's behavior and its applications in various fields.
Electron Configuration and Chemical Behavior
- To truly grasp the periodic table's significance, one must delve into the realm of electron configuration. Each element's position in the table is intricately linked to its electron structure, which, in turn, influences its chemical behavior. Understanding how electrons are arranged within atoms sheds light on why elements in the same group exhibit similar chemical properties while those in the same period differ significantly.
Applications Beyond Chemistry
- While the periodic table is a cornerstone of chemistry, its impact extends far beyond the realm of chemical sciences. Elements play vital roles in diverse fields, including materials science, physics, biology, and even everyday life. For instance, the study of semiconductors and their applications in electronics relies heavily on elements like silicon and germanium. By exploring these interdisciplinary connections, you can appreciate how the periodic table underpins advancements in technology, medicine, and various scientific disciplines.
Recent Discoveries and Superheavy Elements
- The periodic table is not static; it evolves as scientists continue to explore and discover new elements. In recent years, researchers have pushed the boundaries of the table with the synthesis of superheavy elements, such as nihonium (Nh) and tennessine (Ts). Learning about these groundbreaking discoveries and the challenges involved in creating and studying these elements offers a glimpse into the frontiers of modern science.
Theoretical Extensions and Uncharted Territory
- Beyond the known elements, there exist theoretical predictions of even more exotic forms of matter, such as island of stability in superheavy elements. This area of theoretical chemistry and physics explores the possibilities of elements with properties yet to be observed. While these elements may not be part of the periodic table today, they represent the uncharted territory awaiting exploration by future generations of scientists.
By delving into these advanced topics, you can gain a deeper appreciation for the periodic table's role in shaping our understanding of the universe and its ongoing relevance in cutting-edge scientific research.