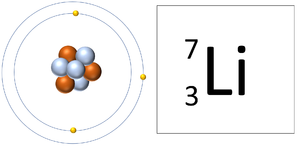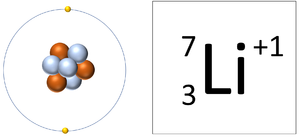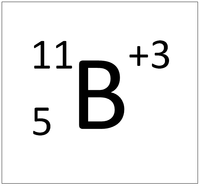Difference between revisions of "Ion"
| Line 67: | Line 67: | ||
Number of [[electron]]s = 10 | Number of [[electron]]s = 10 | ||
|} | |} | ||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782946403/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782946403&linkCode=as2&tag=nrjc-21&linkId=32a0abb60dff015b15b50e9b1d7b4644 ''Ions, page 112, GCSE Combined Science Trilogy; Physics, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945970/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945970&linkCode=as2&tag=nrjc-21&linkId=a120d24dcc7cc7a58192069a3aafc1d2 ''Ions, page 124, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/019835939X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=019835939X&linkCode=as2&tag=nrjc-21&linkId=57e96876985fc39b1a3d8a3e3dc238b6 ''Ions, page 50, GCSE Physics; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Ions, pages 121-2, 148, 339, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Ions, pages 16, 38-43, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294639X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294639X&linkCode=as2&tag=nrjc-21&linkId=51599bb45a2bfaf7c1b6a978b2ca2616 ''Ions, pages 23, 29, 70-74, GCSE Combined Science Trilogy; Chemistry, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945571/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945571&linkCode=as2&tag=nrjc-21&linkId=9e29fad914244909903e5e93f8a01d166 ''Ions, pages 28-30, GCSE Chemistry; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/178294558X/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=178294558X&linkCode=as2&tag=nrjc-21&linkId=f0dfb66dafcb0c6e9449e7b1a4ae1ac266 ''Ions, pages 43, 44, GCSE Physics; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Ions, pages 6-7, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945598/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945598&linkCode=as2&tag=nrjc-21&linkId=ad276ad49df77ab4b40ab4fd0fe10017 ''Ions, pages 96, 113-115, 197, GCSE Combined Science; The Revision Guide, CGP, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851346/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851346&linkCode=as2&tag=nrjc-21&linkId=3ac654f4b0da781c49c855a1af4c92ea ''Ions; charge on, page 35, GCSE Chemistry, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1471851354/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1471851354&linkCode=as2&tag=nrjc-21&linkId=9012a0d354024419214fb3ad5ac44ba0 ''Ions; charge on, pages 150-1, GCSE Combined Science Trilogy 1, Hodder, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Ions; electrolysis, pages 102, 104-105, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Ions; periodicity, pages 40-41, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Ions; pH scale, page 99, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Ions; tests, pages 186-191, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Ions; transition elements, page 33, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
Revision as of 17:35, 7 November 2019
Key Stage 4
Meaning
An Ion is a particle that has a different number of protons to electrons.
About Ions
- An atom contains the same number of protons as electrons. When an atom loses or gains electrons it becomes an ion.
- Ions can be positively charged or negatively charged.
- If an atom gains electrons it becomes a negatively charged ion since there are more electrons than protons and electrons carry a negative charge.
- If an atom loses electrons it becomes a positively charged ion since there are more protons than electrons and protons carry a positive charge.
| Atom | Ion |
| A Lithium atom has 3 protons and 3 electrons so it is neutral. | A Lithium ion has 3 protons and 2 electrons so it has a positive charge. It now has a full Outer Shell. |
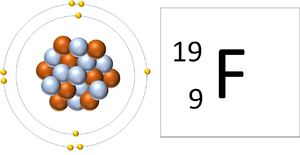
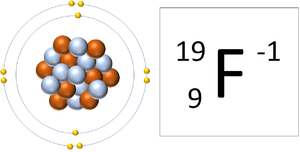
| A Fluorine atom has 9 protons and 9 electrons so it is neutral. | A Fluorine ion has 9 protons and 10 electrons so it is negative charge. It now has a full Outer Shell. |
- The number of electron in an ion can be found using the Atomic Number (which is the same as the Relative Atomic Charge of the nucleus) and subtracting the charge of the ion.
| A Lithium Ion | A Fluorine Ion | A Boron Ion | An Oxygen Ion |
| This ion has an Atomic Number (Z) of 3 and a Relative Atomic Charge (Q) of +1.
Number of electrons = A - Q Number of electrons = 3 - 1 Number of electrons = 2 |
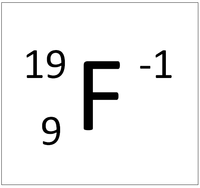
This ion has an Atomic Number (Z) of 9 and a Relative Atomic Charge (Q) of -1.
Number of electrons = A - Q Number of electrons = 9 - (-1) Number of electrons = 10 |
This ion has an Atomic Number (Z) of 5 and a Relative Atomic Charge (Q) of +3.
Number of electrons = A - Q Number of electrons = 5 - 3 Number of electrons = 2 |
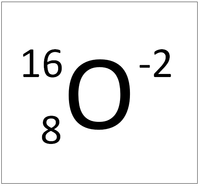
This ion has an Atomic Number (Z) of 8 and a Relative Atomic Charge (Q) of -2.
Number of electrons = A - Q Number of electrons = 8 - (-2) Number of electrons = 10 |
References
AQA
- Ions, page 112, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Ions, page 124, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Ions, page 50, GCSE Physics; Third Edition, Oxford University Press, AQA
- Ions, pages 121-2, 148, 339, GCSE Combined Science Trilogy 1, Hodder, AQA
- Ions, pages 16, 38-43, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Ions, pages 23, 29, 70-74, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Ions, pages 28-30, GCSE Chemistry; The Revision Guide, CGP, AQA
- Ions, pages 43, 44, GCSE Physics; The Revision Guide, CGP, AQA
- Ions, pages 6-7, GCSE Chemistry, Hodder, AQA
- Ions, pages 96, 113-115, 197, GCSE Combined Science; The Revision Guide, CGP, AQA
- Ions; charge on, page 35, GCSE Chemistry, Hodder, AQA
- Ions; charge on, pages 150-1, GCSE Combined Science Trilogy 1, Hodder, AQA
- Ions; electrolysis, pages 102, 104-105, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Ions; periodicity, pages 40-41, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Ions; pH scale, page 99, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Ions; tests, pages 186-191, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Ions; transition elements, page 33, GCSE Chemistry; Third Edition, Oxford University Press, AQA