Difference between revisions of "Transition Metal"
(→Melting Point) |
(→Key Stage 4) |
||
| Line 64: | Line 64: | ||
|- | |- | ||
|[[File:TransitionMetalMeltingPoints.png|center|600px]] | |[[File:TransitionMetalMeltingPoints.png|center|600px]] | ||
| + | | style="height:20px; width:600px; text-align:center;" |N.B. The [[Period 7]] [[element]]s have not been included as they do not occur naturally and have not been made in large enough quantities to find their [[Melting Point|melting points]]. | ||
|} | |} | ||
Revision as of 15:38, 18 December 2018
Contents
Key Stage 4
Meaning
Transition Metals (also known as transition elements) are a block of elements on the Periodic Table between Group 2 and Group 3.
About Transition Metals
| Period 4 | 21Sc | 22Ti | 23V | 24Cr | 25Mn | 26Fe | 27Co | 28Ni | 29Cu | 30Zn |
| Period 5 | 39Y | 40Zr | 41Nb | 42Mo | 43Tc | 44Ru | 45Rh | 46Pd | 47Ag | 48Cd |
| Period 6 | 57La | 72Hf | 73Ta | 74W | 75Re | 76Os | 77Ir | 78Pt | 79Au | 80Hg |
| Period 7 | 89Ac | 104Rf | 105Db | 106Sg | 107Bh | 108Hs | 109Mt | 110Ds | 111Rg | 112Cn |
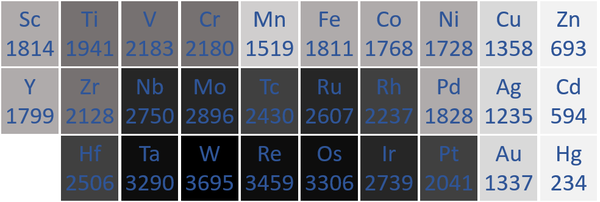
Melting Point
- Transition Metals usually have high melting points.
| The transition metal melting points measured in Kelvin are written below each chemical symbol. | |
| N.B. The Period 7 elements have not been included as they do not occur naturally and have not been made in large enough quantities to find their melting points. |
Density
- Transition Metals have a high density compared to other elements. However, the Lanthanide
Reactivity|Reactivity
Ion Formation
(Colour)