Difference between revisions of "Separating Mixtures"
| (15 intermediate revisions by 2 users not shown) | |||
| Line 6: | Line 6: | ||
: Different [[mixture]]s need to be '''separated''' in different ways: | : Different [[mixture]]s need to be '''separated''' in different ways: | ||
*Sieving | *Sieving | ||
| − | *Filtering | + | *[[Filtering]] |
| − | *Evaporating | + | *[[Evaporation of Solutions|Evaporating]] |
===Examples=== | ===Examples=== | ||
| Line 16: | Line 16: | ||
| style="height:20px; width:200px; text-align:center;" |Sea Water | | style="height:20px; width:200px; text-align:center;" |Sea Water | ||
|- | |- | ||
| + | |[[File:PebblesandSand.png|center|200px]] | ||
| + | |[[File:MuddyPuddle.png|center|200px]] | ||
|[[File:Sea.png|center|200px]] | |[[File:Sea.png|center|200px]] | ||
| − | |||
| − | |||
|- | |- | ||
| style="height:20px; width:200px; text-align:center;" |You can separate the sand from the pebbles with a sieve. | | style="height:20px; width:200px; text-align:center;" |You can separate the sand from the pebbles with a sieve. | ||
| Line 28: | Line 28: | ||
|[[File:EvaporatingDish.png|center|200px]] | |[[File:EvaporatingDish.png|center|200px]] | ||
|} | |} | ||
| + | |||
| + | ==Key Stage 3== | ||
| + | ===Meaning=== | ||
| + | [[Separating Mixtures]] is when you take out one or more of the [[substance]]s from a [[mixture]]. | ||
| + | |||
| + | ===About Separating Mixtures=== | ||
| + | : All [[mixture]]s can be '''separated''' without a [[Chemical Reaction|chemical reaction]]. | ||
| + | : There are several methods of '''separating mixtures''' that you should know: | ||
| + | *[[Filtration]] | ||
| + | *[[Evaporation of Solutions|Evaporation]] | ||
| + | *[[Distillation]] | ||
| + | *[[Chromatography]] | ||
| + | |||
| + | ===Examples=== | ||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |'''Filtration''' | ||
| + | | style="height:20px; width:200px; text-align:center;" |'''Evaporation''' | ||
| + | |- | ||
| + | |[[File:Filtration.png|center|300px]] | ||
| + | |[[File:EvaporatingClipart.png|center|300px]] | ||
| + | |- | ||
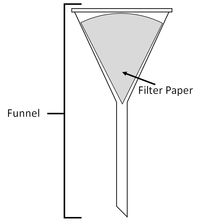
| + | | style="height:20px; width:200px; text-align:center;" |A [[mixture]] or a [[liquid]] and an [[insoluble]] [[solid]] can be '''separated''' by [[filtration]]. | ||
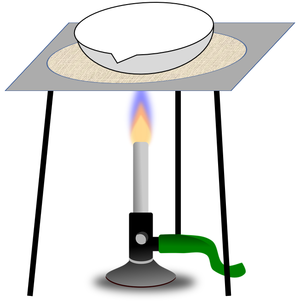
| + | | style="height:20px; width:200px; text-align:center;" |A [[solute]] can be recovered from a [[solution]] by [[evaporating]] away the [[solvent]]. | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |'''Distillation''' | ||
| + | | style="height:20px; width:200px; text-align:center;" |'''Chromatography''' | ||
| + | |- | ||
| + | |[[File:Distillation.png|center|300px]] | ||
| + | |[[File:ChromatographyDiagram.png|center|300px]] | ||
| + | |- | ||
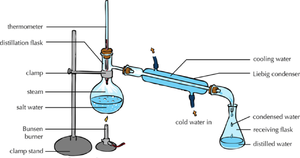
| + | | style="height:20px; width:200px; text-align:center;" |The [[solute]] and [[solvent]] can be '''separated''' by [[distillation]]. | ||
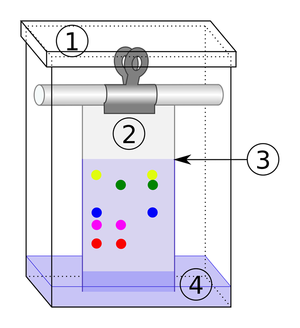
| + | | style="height:20px; width:200px; text-align:center;" |When there are more than 1 [[solute]]s in a [[solution]] [[chromatography]] can '''separate''' them. | ||
| + | |} | ||
| + | |||
| + | |||
| + | ==Key Stage 4== | ||
| + | ===Meaning=== | ||
| + | To '''separate a mixture''' is to remove one or more [[chemical]]s from a [[mixture]] of [[chemical]]s. | ||
| + | |||
| + | ===About Separating Mixtures=== | ||
| + | : Techniques to '''separate mixtures''' are usually used to obtain [[pure]] samples of a [[chemical]]. | ||
| + | Different '''separation''' techniques are needed for different types of [[mixture]]: | ||
| + | *[[Filtration]] - To remove an [[insoluble]] [[solid]] from [[mixture]] with a [[liquid]]. | ||
| + | *[[Evaporation of Solutions|Evaporation]] ([[Evaporation of Solutions|Crystallisation]]) - Collecting a [[solute]] from a [[solution]] while losing the [[solvent]]. | ||
| + | *[[Distillation]] - Collecting both the [[solute]] and [[solvent]] from a [[solution]]. | ||
| + | *[[Fractional Distillation]] - Collecting different [[solvent]]s from the same [[solution]]. | ||
| + | *[[Chromatography]] - '''Separating''' two or more [[solute]]s in [[solution]]. | ||
| + | *[[Centrifuge]] - '''Separating''' [[substance]]s in a [[mixture]] by their [[density]]. | ||
| + | |||
| + | ===Extra Information=== | ||
| + | {{#ev:youtube|https://www.youtube.com/watch?v=q8Ent5CXhfY}} | ||
| + | |||
| + | ===References=== | ||
| + | ====Edexcel==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1782945741/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945741&linkCode=as2&tag=nrjc-21&linkId=30da4f2178da182547b62a7329d13b57 ''Separating mixtures, pages 100-102, GCSE Combined Science; The Revision Guide, CGP, Edexcel ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945725/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945725&linkCode=as2&tag=nrjc-21&linkId=694be7494de75af3349537d34e13f7f0 ''Separating mixtures, pages 37-39, GCSE Chemistry; The Revision Guide, CGP, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Separating mixtures, pages 27-29, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945695/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945695&linkCode=as2&tag=nrjc-21&linkId=ceafcc80bcad6b6754ee97a0c7ceea53 ''Separating mixtures, pages 99-103, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR ''] | ||
Latest revision as of 10:45, 19 December 2019
Contents
Key Stage 2
Meaning
Separating Mixtures is when you take out one of the substances from a mixture.
About Separating Mixtures
- Different mixtures need to be separated in different ways:
- Sieving
- Filtering
- Evaporating
Examples
| Beach | Muddy Puddle | Sea Water |
| You can separate the sand from the pebbles with a sieve. | You can separate mud from water in the puddle with filter paper and a funnel. | You can separate salt from the water by evaporating the water in an evaporating dish. |
Key Stage 3
Meaning
Separating Mixtures is when you take out one or more of the substances from a mixture.
About Separating Mixtures
- All mixtures can be separated without a chemical reaction.
- There are several methods of separating mixtures that you should know:
Examples
| Filtration | Evaporation |
| A mixture or a liquid and an insoluble solid can be separated by filtration. | A solute can be recovered from a solution by evaporating away the solvent. |
| Distillation | Chromatography |
| The solute and solvent can be separated by distillation. | When there are more than 1 solutes in a solution chromatography can separate them. |
Key Stage 4
Meaning
To separate a mixture is to remove one or more chemicals from a mixture of chemicals.
About Separating Mixtures
Different separation techniques are needed for different types of mixture:
- Filtration - To remove an insoluble solid from mixture with a liquid.
- Evaporation (Crystallisation) - Collecting a solute from a solution while losing the solvent.
- Distillation - Collecting both the solute and solvent from a solution.
- Fractional Distillation - Collecting different solvents from the same solution.
- Chromatography - Separating two or more solutes in solution.
- Centrifuge - Separating substances in a mixture by their density.
Extra Information
References
Edexcel
- Separating mixtures, pages 100-102, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Separating mixtures, pages 37-39, GCSE Chemistry; The Revision Guide, CGP, Edexcel