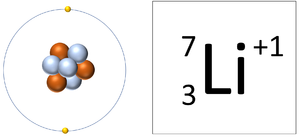Difference between revisions of "Relative Atomic Charge"
(Created page with "==Key Stage 4== ===Meaning=== '''Relative Atomic Charge''' is the charge of a particle compared to the charge of a single proton. ===About Relative Atomic Cha...") |
|||
| (4 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
==Key Stage 4== | ==Key Stage 4== | ||
===Meaning=== | ===Meaning=== | ||
| − | '''Relative Atomic Charge''' is the [[charge]] of a [[particle]] compared to the [[charge]] of a single [[proton]]. | + | '''Relative Atomic Charge''' is the [[Electrical Charge|charge]] of a [[particle]] compared to the [[Electrical Charge|charge]] of a single [[proton]]. |
===About Relative Atomic Charge=== | ===About Relative Atomic Charge=== | ||
| − | : [[Ion]]s, [[Atomic Nucleus|nuclei]] and [[Subatomic Particle|subatomic particles]] have extremely small [[charge]]s such as the [[proton]] (1.6x10<sup>-19</sup>Coulombs) so instead of stating the [[charge]] in [[Coulomb]]s it is compared to the [[charge]] of a [[proton]]. | + | : [[Ion]]s, [[Atomic Nucleus|nuclei]] and [[Subatomic Particle|subatomic particles]] have extremely small [[Electrical Charge|charge]]s such as the [[proton]] (1.6x10<sup>-19</sup>Coulombs) so instead of stating the [[Electrical Charge|charge]] in [[Coulomb]]s it is compared to the [[Electrical Charge|charge]] of a [[proton]]. |
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| Line 11: | Line 11: | ||
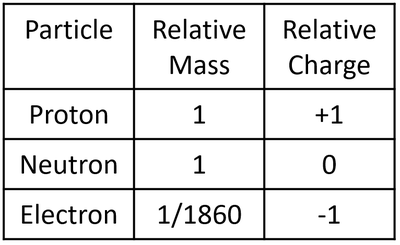
| style="height:20px; width:200px; text-align:center;" |A table showing the [[Relative Atomic Mass|relative mass]] and [[Relative Atomic Charge|relative charge]] of the [[proton]], [[neutron]] and [[electron]]. | | style="height:20px; width:200px; text-align:center;" |A table showing the [[Relative Atomic Mass|relative mass]] and [[Relative Atomic Charge|relative charge]] of the [[proton]], [[neutron]] and [[electron]]. | ||
|} | |} | ||
| − | : The [[charge]] on an [[ion]] is shown at the top right of the [[element]] symbol: | + | : The [[Electrical Charge|charge]] on an [[ion]] is shown at the top right of the [[element]] symbol: |
{| class="wikitable" | {| class="wikitable" | ||
| − | |||
| − | |||
| − | |||
|[[File:LithiumIon+Symbol.png|center|300px]] | |[[File:LithiumIon+Symbol.png|center|300px]] | ||
| + | |[[File:FluorineIon+Symbol.png|center|300px]] | ||
|- | |- | ||
| style="height:20px; width:300px; text-align:center;" |A [[Lithium]] [[ion]] has 3 [[proton]]s and 2 [[electron]]s so it has a [[Positive Charge|positive charge]]. It now has a full [[Outer Shell]]. | | style="height:20px; width:300px; text-align:center;" |A [[Lithium]] [[ion]] has 3 [[proton]]s and 2 [[electron]]s so it has a [[Positive Charge|positive charge]]. It now has a full [[Outer Shell]]. | ||
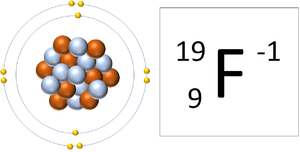
| − | + | | style="height:20px; width:300px; text-align:center;" |A [[Fluorine]] [[ion]] has 9 [[proton]]s and 10 [[electron]]s so it is [[Negative Charge|negative charge]]. It now has a full [[Outer Shell]]. | |
| − | |||
| − | |||
| − | | style="height:20px; width:300px; text-align:center;" |A [[Fluorine]] [[ | ||
|} | |} | ||
Latest revision as of 10:57, 2 February 2019
Key Stage 4
Meaning
Relative Atomic Charge is the charge of a particle compared to the charge of a single proton.
About Relative Atomic Charge
- Ions, nuclei and subatomic particles have extremely small charges such as the proton (1.6x10-19Coulombs) so instead of stating the charge in Coulombs it is compared to the charge of a proton.
| A table showing the relative mass and relative charge of the proton, neutron and electron. |
| A Lithium ion has 3 protons and 2 electrons so it has a positive charge. It now has a full Outer Shell. | A Fluorine ion has 9 protons and 10 electrons so it is negative charge. It now has a full Outer Shell. |