Difference between revisions of "Unstable Isotope"
| Line 17: | Line 17: | ||
|- | |- | ||
|[[File:NuclearKey.png|center|200px]] | |[[File:NuclearKey.png|center|200px]] | ||
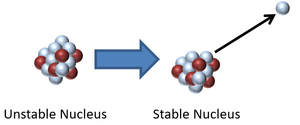
| − | | style="height:20px; width: | + | |[[File:NeutronDecay3.png|center|300px]] |
| + | |- | ||
| + | | style="height:20px; width:300px; text-align:center;" |This is a key to show the types of [[particle]]s in the following [[Radioactive Decay|decays]] of [[Unstable Isotope|unstable nuclei]]. | ||
| + | | style="height:20px; width:300px; text-align:center;" |This [[Atomic Nucleus|nucleus]] is [[Unstable Isotope|unstable]] because it has too many [[neutron]]s relative to [[proton]]s so it [[Radioactive Decay|decays]] via [[Neutron Radiation|neutron radiation]] reducing the [[Relative Atomic Mass|atomic mass]] by 1. | ||
|} | |} | ||
| Line 25: | Line 28: | ||
|[[File:BetaDecay3.png|center|300px]] | |[[File:BetaDecay3.png|center|300px]] | ||
|- | |- | ||
| − | | style="height:20px; width:300px; text-align:center;" |This [[Atomic Nucleus|nucleus]] is [[Unstable Isotope|unstable]] because it is too [[massive]] | + | | style="height:20px; width:300px; text-align:center;" |This [[Atomic Nucleus|nucleus]] is [[Unstable Isotope|unstable]] because it is too [[massive]] and has too few [[neutron]]s relative to [[proton]]s so it [[Radioactive Decay|decays]] via [[Alpha Radiation|alpha emission]] reducing the [[Relative Atomic Mass|atomic mass]] by 4 and the [[Atomic Number|atomic number]] by 2. |
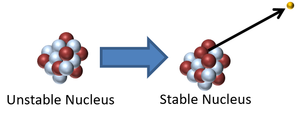
| style="height:20px; width:300px; text-align:center;" |This [[Atomic Nucleus|nucleus]] is [[Unstable Isotope|unstable]] because it is too many [[neutron]]s so it [[Radioactive Decay|decay]]s via [[Beta Radiation|beta emission]] in which a [[neutron]] turns into a [[proton]] increasing the [[Atomic Number|atomic number]] by 1. | | style="height:20px; width:300px; text-align:center;" |This [[Atomic Nucleus|nucleus]] is [[Unstable Isotope|unstable]] because it is too many [[neutron]]s so it [[Radioactive Decay|decay]]s via [[Beta Radiation|beta emission]] in which a [[neutron]] turns into a [[proton]] increasing the [[Atomic Number|atomic number]] by 1. | ||
|} | |} | ||
Revision as of 10:42, 7 March 2019
Key Stage 4
Meaning
Unstable Isotopes are isotopes which radioactively decay to form new stable isotopes of the same element or a different element.
About Unstable Isotopes
- An isotope may be unstable if:
- It has too many neutrons
- It has too few neutrons
- The nucleus is too massive.
- It has excess vibrational energy.
- An unstable isotope may decay causing it to transmute into a new element or a more stable isotope of the same element by releasing a particle from the nucleus.
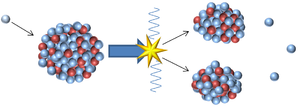
- An unstable isotope may be too massive so it can lose an alpha particle to become a less massive element or it can split into two smaller, more stable elements in a process called nuclear fission.
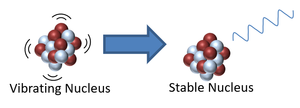
- An unstable isotope may be vibrating with excess energy so to lose this energy it will emit gamma radiation.
| This is a key to show the types of particles in the following decays of unstable nuclei. | This nucleus is unstable because it has too many neutrons relative to protons so it decays via neutron radiation reducing the atomic mass by 1. |
| This nucleus is unstable because it is too massive and has too few neutrons relative to protons so it decays via alpha emission reducing the atomic mass by 4 and the atomic number by 2. | This nucleus is unstable because it is too many neutrons so it decays via beta emission in which a neutron turns into a proton increasing the atomic number by 1. |
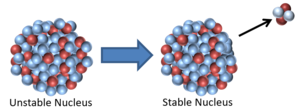
| This nucleus is unstable because it is has excess vibrational energy so it decays by emitting a gamma ray. After the decay it still has the same atomic mass and atomic number but is no longer vibrating. | This nucleus is unstable because it is far too massive and has too many neutrons so it decays by splitting into two smaller, more stable elements and releases some neutrons. |