Difference between revisions of "Fractional Distillation"
(→About Fractional Distillation) |
(→About Fractional Distillation) |
||
| Line 10: | Line 10: | ||
*[[Separating Mixtures|Separating]] two [[solute]]s from each other in [[solution]] - [[Chromatography]] | *[[Separating Mixtures|Separating]] two [[solute]]s from each other in [[solution]] - [[Chromatography]] | ||
*[[Separating Mixtures|Separating]] an [[insoluble]] [[solid]] from a [[soluble]] [[solid]] - [[Filtration]] | *[[Separating Mixtures|Separating]] an [[insoluble]] [[solid]] from a [[soluble]] [[solid]] - [[Filtration]] | ||
| − | + | ===About the Fractional Distillation of Crude Oil=== | |
| + | : [[Crude Oil]] is [[Separating Mixtures|separated]] using '''fractional distillation'''. | ||
| + | : '''Fractional distillation''' is used to separate the different '''fractions''' of [[Crude Oil|crude oil]] which each have different [[Boiling Point|boiling points]]. | ||
: '''Fractional Distillation''' relies on the different [[Boiling Point|boiling points]] of [[liquid]]s. By [[heating]] all [[liquid]]s beyond their [[Boiling Point]] they can be turned into a [[gas]]. Each [[gas]] can then be cooled and [[condensing|condensed]] in tubes kept just below the [[Boiling Point|boiling point]] of each fraction. | : '''Fractional Distillation''' relies on the different [[Boiling Point|boiling points]] of [[liquid]]s. By [[heating]] all [[liquid]]s beyond their [[Boiling Point]] they can be turned into a [[gas]]. Each [[gas]] can then be cooled and [[condensing|condensed]] in tubes kept just below the [[Boiling Point|boiling point]] of each fraction. | ||
| − | : [[Crude Oil]] | + | : The '''fractions''' of [[Crude Oil|crude oil]] separated during '''fractional distillation''' are: |
| + | *[[Natural Gas]] | ||
| + | *LPG | ||
| + | *Gasoline | ||
| + | *Diesel | ||
| + | *Kerosene | ||
| + | *Fuel Oil | ||
| + | *Lubricating Oil | ||
| + | *Bitumen | ||
Revision as of 21:08, 22 September 2019
Contents
Key Stage 4
Meaning
Fractional distillation is a technique which can be used to separate two or more solvents from solution.
About Fractional Distillation
Fractional Distillation can be used for:
- Separating a solution to recover multiple solvents.
Fractional Distillation cannot be used for:
- Separating two solutes from each other in solution - Chromatography
- Separating an insoluble solid from a soluble solid - Filtration
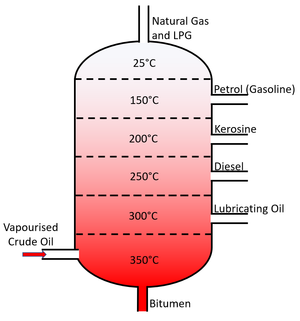
About the Fractional Distillation of Crude Oil
- Crude Oil is separated using fractional distillation.
- Fractional distillation is used to separate the different fractions of crude oil which each have different boiling points.
- Fractional Distillation relies on the different boiling points of liquids. By heating all liquids beyond their Boiling Point they can be turned into a gas. Each gas can then be cooled and condensed in tubes kept just below the boiling point of each fraction.
- The fractions of crude oil separated during fractional distillation are:
- Natural Gas
- LPG
- Gasoline
- Diesel
- Kerosene
- Fuel Oil
- Lubricating Oil
- Bitumen