Difference between revisions of "Chemical Bond"
(→Examples) |
|||
| Line 10: | Line 10: | ||
===Examples=== | ===Examples=== | ||
| + | {| class="wikitable" | ||
| + | |+ Covalent Bonds | ||
| + | |- | ||
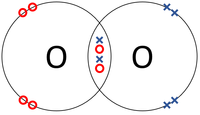
| + | |[[File:OxygenDotandCrossDiagram.png|center|200px]] | ||
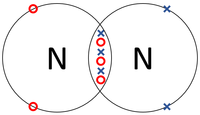
| + | |[[File:NitrogenDotandCrossDiagram.png|center|200px]] | ||
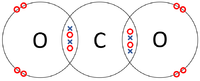
| + | |[[File:CarbonDioxideDotandCrossDiagram.png|center|200px]] | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |+ Ionic Bonds | ||
| + | |- | ||
| + | |[[File:LithiumFluorideDotandCross.png|center|200px]] | ||
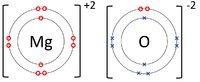
| + | |[[File:MagnesiumOxideDotandCrossDiagram.png|center|200px]] | ||
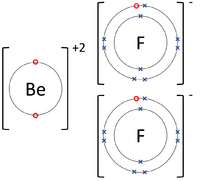
| + | |[[File:BerylliumFluorideDotandCrossDiagram.png|center|200px]] | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | |+ Ionic Bonds | ||
| + | |- | ||
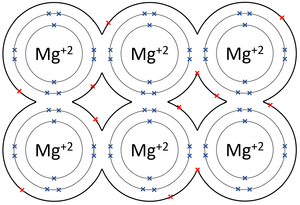
| + | |[[File:MagnesiumMetallicBond.png|center|300px]] | ||
| + | |[[File:AluminiumDotandCrossDiagram.png|center|300px]] | ||
| + | |- | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | | style="height:20px; width:200px; text-align:center;" |Text | ||
| + | |} | ||
Revision as of 16:35, 20 December 2018
Key Stage 4
Meaning
A chemical bond is a force of attraction holding the atoms inside a molecule together.
About Chemical Bonds
There are three types of chemical bond you should know:
- Covalent Bonds - In which atoms share electrons with one another.
- Ionic Bonds - In which electrons are transferred from one atom to another.
- Metallic Bonds - In which some electrons move freely between atoms creating lattice of positively charged ions surrounded by a sea of delocalised electrons (free electrons).
Examples
| Text | Text | Text |
| Text | Text | Text |
| Text | Text |