Difference between revisions of "Diamond"
(Created page with "==Key Stage 3== ===Meaning=== Diamonds are large crystal molecules of carbon. ===About Diamonds=== : Diamonds are very hard. : Diamonds a...") |
|||
| Line 22: | Line 22: | ||
: [[Diamond]]s have a high [[Melting Point|melting point]]. | : [[Diamond]]s have a high [[Melting Point|melting point]]. | ||
: [[Diamond]]s are [[transparent]] and have a high [[Refractive Index|refractive index]]. | : [[Diamond]]s are [[transparent]] and have a high [[Refractive Index|refractive index]]. | ||
| + | : [[Diamond]] is a poor [[Electrical Conductor|electrical conductor]] because there are no free [[electron]]s to move around the [[Giant Covalent Structure|giant covalent structure]]. | ||
Revision as of 19:33, 31 December 2018
Key Stage 3
Meaning
Diamonds are large crystal molecules of carbon.
About Diamonds
- Diamonds are very hard.
- Diamonds are transparent.
Key Stage 4
Meaning
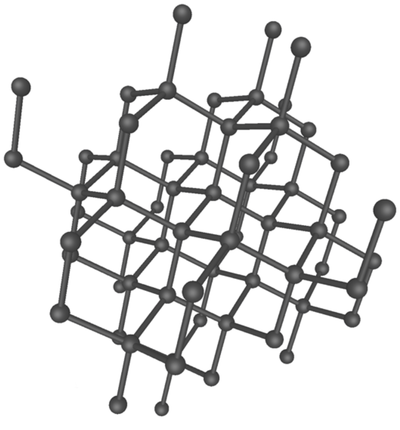
Diamond is an allotrope of carbon in which the atoms are bonded in a tetrahedral arrangement.
| A diagram showing the arrangement of carbon atoms in diamond. |
About Diamonds
- Diamonds are a giant covalent structure.
- Diamonds are extremely hard.
- Diamonds have a high melting point.
- Diamonds are transparent and have a high refractive index.
- Diamond is a poor electrical conductor because there are no free electrons to move around the giant covalent structure.