Key Stage 4
Meaning
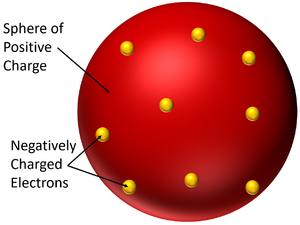
The Plum Pudding Model is a model of the atom which suggests the atom is a solid sphere of positive charge with negatively charged electrons spread within it.
About the Plum Pudding Model
- In the Plum Pudding Model the atom is neutral because the negatively charged electrons are fixed within a larger sphere of positive charge.
- The Plum Pudding Model is named after a desert made from sponge with plums stuck inside. It was imagined that the atom had a sphere of positive charge like the sponge of the cake and electrons suck inside, like the plums stuck inside the sponge.
- The Plum Pudding Model was proposed by J.J. Thompson who discovered the electron and realised it was part of an atom.
- The Plum Pudding Model was proven false by Rutherford's Alpha Scattering Experiment and was replaced by the Nuclear Model.