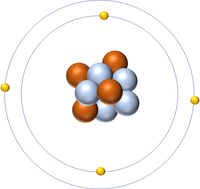
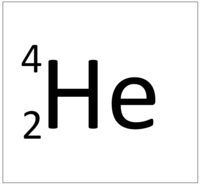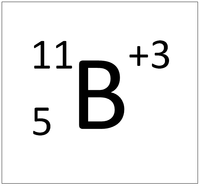Calculating the Numbers of Protons, Neutrons and Electrons
Key Stage 4
Meaning
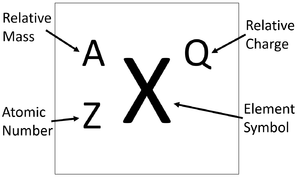
Calculting the numbers of protons, neutrons, and electrons can be done from information about the Atomic Number, Relative Atomic Mass and Relative Atomic Charge of a particle.
About Calculating the Numbers of Protons, Neutrons and Electrons
- The number of protons in an atom or ion is given by the Atomic Number.
| Hydrogen | Helium | Lithium | Beryllium |
| Hydrogen has an Atomic Number of 1 so it has 1 proton. | Helium has an Atomic Number of 2 so it has 2 protons. | Lithium has an Atomic Number of 3 so it has 3 protons. | Beryllium has an Atomic Number of 4 so it has 4 protons. |
- The number of neutrons can be found by subtracting the Atomic Number from the Relative Atomic Mass.
| Hydrogen | Helium | Lithium | Beryllium |
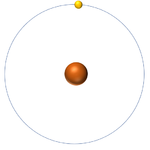
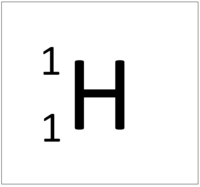
| This atom has an Atomic Number (Z) of 1 and a Relative Atomic Mass (A) of 1.
Number of neutrons = A - Z Number of neutrons = 1 - 1 Number of neutrons = 0 |
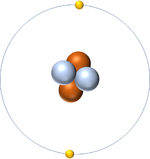
This atom has an Atomic Number (Z) of 2 and a Relative Atomic Mass (A) of 4.
Number of neutrons = A - Z Number of neutrons = 4 - 2 Number of neutrons = 2 |
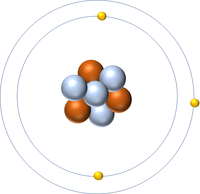
This atom has an Atomic Number (Z) of 3 and a Relative Atomic Mass (A) of 7.
Number of neutrons = A - Z Number of neutrons = 7 - 3 Number of neutrons = 4 |
This atom has an Atomic Number (Z) of 4 and a Relative Atomic Mass (A) of 9.
Number of neutrons = A - Z Number of neutrons = 9 - 4 Number of neutrons = 5 |
- The number of electron in an ion can be found using the Atomic Number (which is the same as the Relative Atomic Charge of the nucleus) and subtracting the charge of the ion.
| A Lithium Ion | A Fluorine Ion | A Boron Ion | An Oxygen Ion |
| This ion has an Atomic Number (Z) of 3 and a Relative Atomic Charge (Q) of +1.
Number of electrons = A - Q Number of electrons = 3 - 1 Number of electrons = 2 |
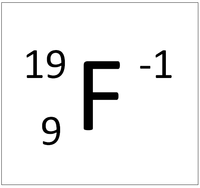
This ion has an Atomic Number (Z) of 9 and a Relative Atomic Charge (Q) of -1.
Number of electrons = A - Q Number of electrons = 9 - (-1) Number of electrons = 10 |
This ion has an Atomic Number (Z) of 5 and a Relative Atomic Charge (Q) of +3.
Number of electrons = A - Q Number of electrons = 5 - 3 Number of electrons = 2 |
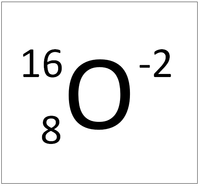
This ion has an Atomic Number (Z) of 8 and a Relative Atomic Charge (Q) of -2.
Number of electrons = A - Q Number of electrons = 8 - (-2) Number of electrons = 10 |