Negative Ion
Contents
Key Stage 4
Meaning
Negative ions are elements or compounds which have gained one or more electrons to become negatively charged.
About Negative Ions
- In chemical reactions between metals and non-metals the non-metal elements form negative ions.
- In alkalis there are free moving hydroxide ions which are negative ions. The concentration of these OH- ions is what determines the alkalinity of a solution.
- Negative ions are attracted to positive ions and to the positive electrode (anode) during electrolysis.
Examples
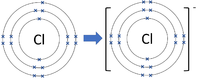
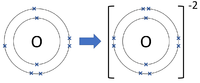
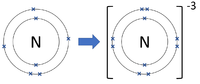
| Chlorine forms -1 ions. | Oxygen forms -2 ions. | Nitrogen forms -3 ions. |
References
AQA
- Negative ion, testing, page 263, GCSE Chemistry; Student Book, Collins, AQA
- Negative ions, page 70, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Negative ions, pages 39, 188-189, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Negative ions, pages 72, 258-261, GCSE Chemistry, CGP, AQA