Difference between revisions of "Giant Ionic Structure"
| Line 12: | Line 12: | ||
|[[File:GiantIonicSodiumChloride.png|center|400px]] | |[[File:GiantIonicSodiumChloride.png|center|400px]] | ||
|- | |- | ||
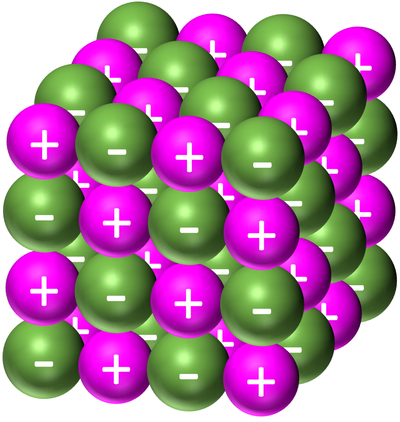
| − | | style="height:20px; width:200px; text-align:center;" |[[Sodium Chloride]] forms a '''giant ionic structure'''. The [[Sodium]] is represented by the light | + | | style="height:20px; width:200px; text-align:center;" |[[Sodium Chloride]] forms a '''giant ionic structure'''. The [[Sodium]] is represented by the light purple [[Positive Ion|positive ion]]s and the [[Chloride]] is represented by the green [[Negative Ion|negative ions]]. |
|} | |} | ||
Revision as of 18:56, 8 April 2019
Contents
Key Stage 4
Meaning
Giant ionic structures are large molecules in which the atoms are held together by ionic bonds.
About Giant Ionic Structures
- Giant ionic structures are molecules made of a large number of metal and non-metal ions joined by ionic bonds.
- The ions in a giant ionic structure are arranged in a regular lattice (a repeating pattern of elements.
Examples
| Sodium Chloride forms a giant ionic structure. The Sodium is represented by the light purple positive ions and the Chloride is represented by the green negative ions. |
Bulk Properties
- Giant ionic structures are poor electrical conductors because the ions are not free to move.
- Most giant ionic structures can be broken down and dissolved in water.
- Giant ionic structures have high melting points due to the strong electrostatic force between the ions.