Difference between revisions of "Reactivity"
(Created page with "==Key Stage 4== ===Meaning=== Reactivity is how vigorously a chemical will react. ===About Reactivity=== : Reactivity is determined by how e...") |
|||
| (14 intermediate revisions by 2 users not shown) | |||
| Line 5: | Line 5: | ||
===About Reactivity=== | ===About Reactivity=== | ||
: [[Reactivity]] is determined by how easily an [[element]] can lose or gain [[electron]]s. | : [[Reactivity]] is determined by how easily an [[element]] can lose or gain [[electron]]s. | ||
| + | : [[Electron]]s are held in [[orbit]] around the [[Atomic Nucleus|nucleus]] because the [[electron]]s are [[Negative Charge|negatively charged]] and are attracted to the [[Atomic Nucleus|nucleus]] which is [[Positive Charge|positively charged]]. | ||
| + | : If an [[element]] loses [[electron]]s easily it is highly '''reactive'''. | ||
| + | : If an [[element]] gains [[electron]]s readily it is also highly '''reactive'''. | ||
| + | |||
Three important factors affect [[reactivity]] of [[element]]s. | Three important factors affect [[reactivity]] of [[element]]s. | ||
| − | *The [[charge]] of the [[Atomic Nucleus|nucleus]] | + | *The [[Electrical Charge|charge]] of the [[Atomic Nucleus|nucleus]] |
*The shielding effect of inner [[electron]]s. | *The shielding effect of inner [[electron]]s. | ||
*Distance between the [[Atomic Nucleus|nucleus]] and the [[Outer Shell|outer shell]]. | *Distance between the [[Atomic Nucleus|nucleus]] and the [[Outer Shell|outer shell]]. | ||
| + | |||
| + | ====Reactivity in Groups 1, 2 and 3==== | ||
| + | {| class="wikitable" | ||
| + | |- | ||
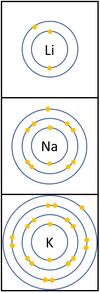
| + | |[[File:Group1ElectronShells.png|center|100px]] | ||
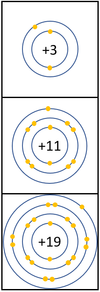
| + | |[[File:Group1ElectronShielding.png|center|100px]] | ||
| + | | style="height:20px; width:400px; text-align:left;" |In a [[Chemical Reaction|chemical reaction]] the [[electron]] in the [[Outer Shell|outer shell]] is lost. | ||
| + | |||
| + | The [[reactivity]] increases as you go down the group because: | ||
| + | *The outer [[electron]] is further away from the [[Atomic Nucleus|nucleus]] with each additional [[Electron Orbital|electron shell]] making the [[force]] of [[attract]]ion weaker. This makes it easier for an [[atom]] to lose it's outer [[electron]]. | ||
| + | *Even though the [[Electrical Charge|charge]] of the [[Atomic Nucleus|nucleus]] increases the outer [[electron]] is shielded from most of the [[Positive Charge|positive charge]] of the [[Atomic Nucleus|nucleus]] by [[electron]]s in the inner shells. | ||
| + | |} | ||
| + | |||
| + | ====Reactivity in Group 7==== | ||
| + | {| class="wikitable" | ||
| + | |- | ||
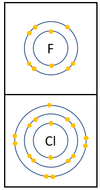
| + | |[[File:Group7ElectronShells.png|center|100px]] | ||
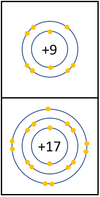
| + | |[[File:Group7ElectronShielding.png|center|100px]] | ||
| + | | style="height:20px; width:400px; text-align:left;" |In a [[Chemical Reaction|chemical reaction]] an extra [[electron]] is added to the [[Outer Shell|outer shell]]. | ||
| + | |||
| + | The [[reactivity]] decreases as you go down the group because: | ||
| + | *The outer [[electron]]s are further away from the [[Atomic Nucleus|nucleus]] with each additional [[Electron Orbital|electron shell]] making the [[force]] of [[attract]]ion weaker. This makes it less able to gain an extra [[electron]]. | ||
| + | *Even though the [[Electrical Charge|charge]] of the [[Atomic Nucleus|nucleus]] increases the outer [[electron]]s are shielded from most of the [[Positive Charge|positive charge]] of the [[Atomic Nucleus|nucleus]] by [[electron]]s in the inner shells. | ||
| + | |} | ||
| + | |||
| + | ====Reactivity along Period 2==== | ||
| + | {| class="wikitable" | ||
| + | |- | ||
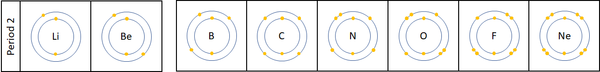
| + | |[[File:Period2ElectronShells.png|center|600px]] | ||
| + | |- | ||
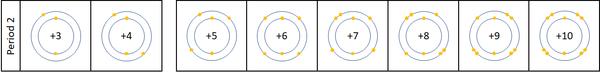
| + | |[[File:Period2ElectronShielding.png|center|600px]] | ||
| + | |- | ||
| + | | style="height:20px; width:600px; text-align:left;" |For the first 3 [[element]]s [[Lithium]], [[Beryllium]] and [[Boron]] all lose [[electron]]s in [[Chemical Reaction|chemical reactions]]. | ||
| + | |||
| + | The [[reactivity]] decreases as you go across the [[Period (Chemistry)|period]] because: | ||
| + | *The outer [[electron]]s are all roughly the same distance away from the [[Atomic Nucleus|nucleus]]. | ||
| + | *The [[Electrical Charge|charge]] on the [[Atomic Nucleus|atomic nucleus]] increases as you move go across the [[period]] but the [[electron]] shielding caused by the two inner [[electron]]s remains the same. This causes the [[electron]]s to experience a greater [[force]] of [[attraction]] as you move along the [[period]], making it harder for the [[atom]]s to lose [[electron]]s and become [[ion]]s. | ||
| + | |||
| + | [[Nitrogen]], [[Oxygen]] and [[Fluorine]] can all gain [[electron]]s to become [[Negative Charge|negative]] [[ion]]s in certain [[Chemical Reaction|reaction]]s. | ||
| + | |||
| + | The [[reactivity]] increases as you go across the [[Period (Chemistry)|period]] because: | ||
| + | *The outer [[electron]]s are all roughly the same distance away from the [[Atomic Nucleus|nucleus]]. | ||
| + | *The [[Electrical Charge|charge]] on the [[Atomic Nucleus|atomic nucleus]] increases as you move go across the [[period]] but the [[electron]] shielding caused by the two inner [[electron]]s remains the same. This causes the [[electron]]s to experience a greater [[force]] of [[attraction]] as you move along the [[period]], making it easier for an [[atom]]s to gain more [[electron]]s to become [[ion]]s. | ||
| + | |} | ||
| + | |||
| + | |||
| + | ===References=== | ||
| + | ====AQA==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Reactivity; alkali metals, pages 26-27, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Reactivity; metals, pages 26-27, 84-89, 220-221, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359381/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359381&linkCode=as2&tag=nrjc-21&linkId=47c8d1ae58d8b3a5e2094cd447154558 ''Reactivity; periodicity, pages 19, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA ''] | ||
| + | |||
| + | ====Edexcel==== | ||
| + | |||
| + | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Reactivity, pages 86-87, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Reactivity; metals, pages 70-71, 125, 133, 141-143, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Reactivity; noble gases, page 137, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Reactivity; practical activities, pages 264-265, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Reactivity; trends in Periodic Table, pages 133, 135, 137, 142-143, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
Latest revision as of 17:46, 18 December 2019
Contents
Key Stage 4
Meaning
Reactivity is how vigorously a chemical will react.
About Reactivity
- Reactivity is determined by how easily an element can lose or gain electrons.
- Electrons are held in orbit around the nucleus because the electrons are negatively charged and are attracted to the nucleus which is positively charged.
- If an element loses electrons easily it is highly reactive.
- If an element gains electrons readily it is also highly reactive.
Three important factors affect reactivity of elements.
- The charge of the nucleus
- The shielding effect of inner electrons.
- Distance between the nucleus and the outer shell.
Reactivity in Groups 1, 2 and 3
| In a chemical reaction the electron in the outer shell is lost.
The reactivity increases as you go down the group because:
|
Reactivity in Group 7
| In a chemical reaction an extra electron is added to the outer shell.
The reactivity decreases as you go down the group because:
|
Reactivity along Period 2
| For the first 3 elements Lithium, Beryllium and Boron all lose electrons in chemical reactions.
The reactivity decreases as you go across the period because:
Nitrogen, Oxygen and Fluorine can all gain electrons to become negative ions in certain reactions. The reactivity increases as you go across the period because:
|
References
AQA
- Reactivity; alkali metals, pages 26-27, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Reactivity; metals, pages 26-27, 84-89, 220-221, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Reactivity; periodicity, pages 19, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA
Edexcel
OCR
- Reactivity; metals, pages 70-71, 125, 133, 141-143, Gateway GCSE Chemistry, Oxford, OCR
- Reactivity; noble gases, page 137, Gateway GCSE Chemistry, Oxford, OCR
- Reactivity; practical activities, pages 264-265, Gateway GCSE Chemistry, Oxford, OCR
- Reactivity; trends in Periodic Table, pages 133, 135, 137, 142-143, Gateway GCSE Chemistry, Oxford, OCR