Difference between revisions of "Giant Covalent Structure"
| Line 8: | Line 8: | ||
===Examples=== | ===Examples=== | ||
| + | {| class="wikitable" | ||
| + | |- | ||
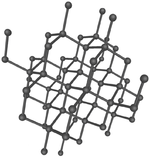
| + | |[[File:DiamondStructure.png|center|150px]] | ||
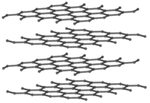
| + | |[[File:GraphiteStructure.png|center|150px]] | ||
| + | |[[File:GrapheneStructure.png|center|150px]] | ||
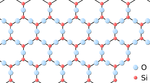
| + | |[[File:SilicaStructure.png|center|150px]] | ||
| + | |- | ||
| + | | style="height:20px; width:150px; text-align:center;" |Text | ||
| + | | style="height:20px; width:150px; text-align:center;" |Text | ||
| + | | style="height:20px; width:150px; text-align:center;" |Text | ||
| + | | style="height:20px; width:150px; text-align:center;" |Text | ||
| + | |} | ||
Revision as of 10:44, 28 December 2018
Contents
Key Stage 4
Meaning
Giant covalent structures are very large molecules in which all the atoms are held to one another by covalent bonds.
About Giant Covalent Structures
- Giant covalent structures are molecules are made of a large number of non-metal atoms joined by covalent bonds.
- The atoms in a giant covalent structure are arranged in a regular lattice (a repeating pattern of elements.
Examples
| Text | Text | Text | Text |
Bulk Properties
- Giant Covalent Structures usually have very high melting points due to the strong chemical bonds between adjacent atoms.
- Giant Covalent Structures may have good or poor electrical conductors depending on whether there are any electrons free to move between atoms.
- Diamond is a poor electrical conductor because all the electrons are shared by adjacent atoms so none are free to move between atoms.
- Graphite and graphene are good electrical conductors because the Carbon atoms only form 3 bonds with adjacent atoms allowing a spare electron in each atom to move freely around the molecule.