Contents
Key Stage 4
Meaning
A dot and cross diagram is a diagram used to show how electrons from the outer shells of atoms are shared or transferred in a chemical bond.
About Dot and Cross Diagrams
- Dot and cross diagrams can be used to represent covalent bonds and ionic bonds.
Examples
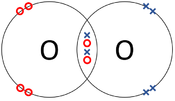
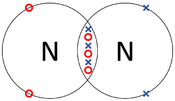
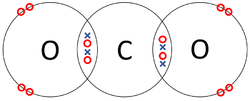
| The two Oxygen atoms each share two of their electrons with one another. | The two Nitrogen atoms each share three of their electrons with one another. | Each Oxygen shares two of its electrons with the Carbon atom while the Carbon atom shares two electrons with each Oxygen atom. |
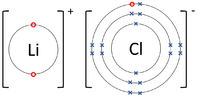
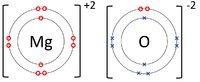
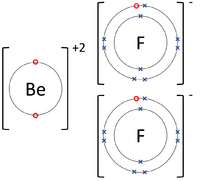
| The Lithium atom donates an electron from its outer shell to the outer shell of the Fluorine atom. | The Magnesium atom donates two electrons from its outer shell to the outer shell of the Oxygen atom. | The Beryllium atom donates two electrons from its outer shell to the outer shells of each Fluorine atom. |
References
AQA
- Dot and cross diagram, pages 43, 60-1, 64-5, 133-5, GCSE Chemistry; Student Book, Collins, AQA
- Dot and cross diagrams, pages 114, 116, 117, GCSE Combined Science; The Revision Guide, CGP, AQA
- Dot and cross diagrams, pages 29, 31, 32, GCSE Chemistry; The Revision Guide, CGP, AQA
- Dot and cross diagrams, pages 39, 44-45, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Dot and cross diagrams, pages 72-76, 78, 79, 81-83, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Dot and cross diagrams, pages 74-78, 80, 81, 83-85, GCSE Chemistry, CGP, AQA
Edexcel
- Dot and cross diagrams, page 184, GCSE Combined Science, Pearson Edexcel
- Dot and cross diagrams, page 40, GCSE Chemistry, Pearson, Edexcel
- Dot and cross diagrams, pages 21, 23, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Dot and cross diagrams, pages 51-53, 56, 58-60, GCSE Chemistry, CGP, Edexcel
- Dot and cross diagrams, pages 84, 86, GCSE Combined Science; The Revision Guide, CGP, Edexcel