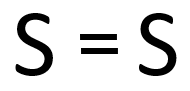Difference between revisions of "Double Bond"
| Line 31: | Line 31: | ||
:[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Double bonds, page 184, GCSE Combined Science, Pearson Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120193/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120193&linkCode=as2&tag=nrjc-21&linkId=572df39392fb4200db8391d98ae6314e ''Double bonds, page 184, GCSE Combined Science, Pearson Edexcel ''] | ||
:[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Double bonds, page 40, GCSE Chemistry, Pearson, Edexcel ''] | :[https://www.amazon.co.uk/gp/product/1292120215/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1292120215&linkCode=as2&tag=nrjc-21&linkId=8f96ddb76196848bafdb124354e4cf77 ''Double bonds, page 40, GCSE Chemistry, Pearson, Edexcel ''] | ||
| + | |||
| + | ====OCR==== | ||
| + | :[https://www.amazon.co.uk/gp/product/0198359829/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=0198359829&linkCode=as2&tag=nrjc-21&linkId=90e8d7b4f039d53035238fa0320fe00b ''Double bonds, page 232, Gateway GCSE Chemistry, Oxford, OCR ''] | ||
| + | :[https://www.amazon.co.uk/gp/product/1782945679/ref=as_li_tl?ie=UTF8&camp=1634&creative=6738&creativeASIN=1782945679&linkCode=as2&tag=nrjc-21&linkId=a2db42f7b4bdf10cafaafa3bb9120940 ''Double bonds, pages 88, 90, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR ''] | ||
Latest revision as of 00:57, 5 December 2019
Contents
Key Stage 4
Meaning
A double bond is a chemical bond in which two electrons are shared or transferred from the outer shell between two atoms.
About Double Bonds
- In covalent bonds a double bond means two electrons from the outer shell of an atom are shared with another atom.
- In ionic bonds a double bond means one of the elements has gained or lost two electrons from/to one other element.
Examples
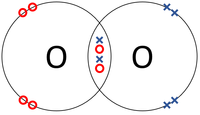
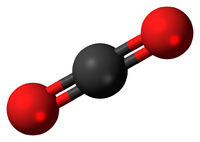
| In this structural diagram a double bond is shown between two Sulphur atoms. | In this dot and cross diagram the two Oxygen atoms in an Oxygen molecule are shown to each share two electrons in a double bond. | In this ball and stick model of Carbon Dioxide the Carbon is shown to share two electron (shown by the two sticks) with each Oxygen atom forming two double bonds. |
References
AQA
- Double bonds, pages 154-155, 158-159, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Double bonds, pages 230-237, GCSE Chemistry, CGP, AQA
- Double bonds, pages 78-80, GCSE Chemistry; The Revision Guide, CGP, AQA
Edexcel
- Double bonds, page 184, GCSE Combined Science, Pearson Edexcel
- Double bonds, page 40, GCSE Chemistry, Pearson, Edexcel