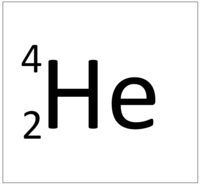Difference between revisions of "Helium"
| Line 33: | Line 33: | ||
| style="height:20px; width:200px; text-align:center;" colspan = "2"|[[Helium]] always has 2 [[proton]]s. The most [[Stable Isotope|stable]] [[isotope]] has 2 [[neutron]]s. | | style="height:20px; width:200px; text-align:center;" colspan = "2"|[[Helium]] always has 2 [[proton]]s. The most [[Stable Isotope|stable]] [[isotope]] has 2 [[neutron]]s. | ||
|} | |} | ||
| + | |||
| + | ==Beyond the Curriculum== | ||
| + | {{#ev:youtube|https://www.youtube.com/watch?v=hLUcO26Q7wE}} | ||
Revision as of 07:58, 7 May 2019
Contents
Key Stage 2
Meaning
Key Stage 3
Meaning
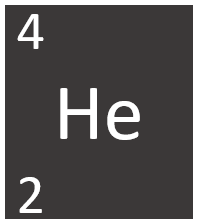
Helium is a Group 0 element, on the Periodic Table, with an atomic number of 2.
About Helium
- Helium has the chemical formula He.
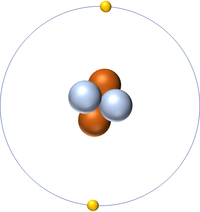
- Helium has two protons and two neutrons in its nucleus giving it an Atomic Number of 2 and an atomic mass of 4.
- Helium is a Noble Gas.
- Helium is a gas at room temperature.
- Helium gas is less dense than air.
- An atom of Helium has a full outer shell of two electrons so it is inert.
Key Stage 4
Meaning
Helium is a Group 0 element, on the Periodic Table, with 2 protons in the nucleus.
About Helium
- Helium has the chemical formula He.
- The most stable isotope of Helium has two neutrons in its nucleus giving it an atomic mass of 4.
- Helium is a Noble Gas.
- Helium is a gas at standard temperature and pressure.
- Helium gas is less dense than air.
- An atom of Helium has a full outer shell of two electrons so it is inert.
| Helium always has 2 protons. The most stable isotope has 2 neutrons. | |