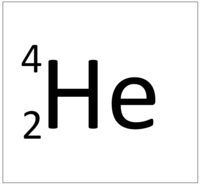Relative Atomic Mass
Revision as of 11:41, 30 September 2018 by NRJC (talk | contribs) (Created page with "==Key Stage 3== ===Meaning=== right|400px|thumb|An [[element tile showing the '''mass number'''.]] The Atomic Mass or '''mass number''' is the num...")
Key Stage 3
Meaning
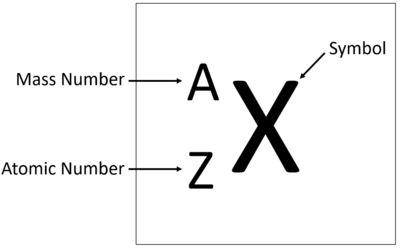
An element tile showing the mass number.
The Atomic Mass or mass number is the number of nucleons (protons + neutrons) in an atom.
About The Atomic Mass
- Two atoms of the same element may have the same Atomic Number but a different Atomic Mass depending on the number of neutrons in the nucleus. Elements with different mass numbers are called isotopes.
- The atomic mass is not affected by the number of electrons.
- Only the particles in the nucleus affect the atomic mass.
Examples
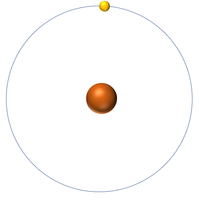
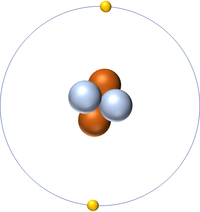
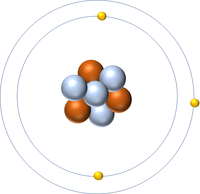
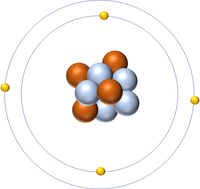
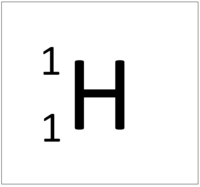
| Hydrogen | Helium | Lithium | Beryllium |
| Hydrogen has one nucleon so it has an atomic mass of 1. | Helium has four nucleons so it has an atomic mass of 4. | Lithium has seven nucleons so it has an atomic mass of 7. | Beryllium has eleven nucleons so it has an atomic mass of 11. |