Extracting Metals by Electrolysis
Contents
Key Stage 3
Meaning
Electrolysis is a way to extract metals from minerals when the metal is more reactive than Carbon.
About Extracting Metals by Electrolysis
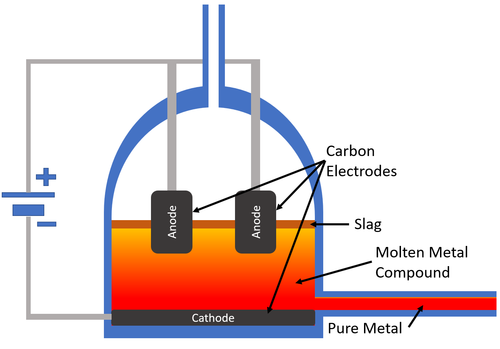
- Metals that are more reactive than Carbon cannot be displaced with Carbon so electrolysis is used to extract them.
| Carbon electrodes are used to pass an Electrical Current through the molten mineral. The metal collects at the cathode which carries a negative charge and the pure metal is removed from the electrolysis cell. |
Key Stage 4
Meaning
Extraction of metals by electrolysis is a method of obtaining a metal from a mineral.
About Extracting Metals by Electrolysis
- Electrolysis is usually used to extract metals when the metal is more reactive than Carbon or the mineral is soluble in water making it cheaper to extract by electrolysis than by smelting.
There are two possible methods used to extract metals by electrolysis:
- Electrolysis of the Mineral in Solution - Used when the mineral is soluble in water and the metal does not react with water.
- Electrolysis of the Molten Mineral - Used when the mineral is insoluble in water or the metal reacts strongly with water.
- Aluminium is a special case in which the mineral is not dissolved in water but in a solvent called Cryolite at very high temperatures. This is done because the mineral melts at an even higher temperature than the cryolite so it is easier to dissolve it in the cryolite than to heat the mineral beyond its melting point.
Electrolysis of Molten Minerals
Used when the mineral is insoluble in water or the metal reacts strongly with water.
- The electrolysis of molten ionic compounds decomposes those ionic compounds in a liquid state.
- The ionic compound must be heated and melted before electrolysis.
- Non-metal ionic elements or compounds will be collected at the anode where they lose their extra electrons.
- Metal ions will be collected at the cathode where they gain electrons.
Electrolysis of Aqueous Minerals
Used when the mineral is soluble in water and the metal does not react with water.
- The electrolysis of aqueous ionic compounds decomposes those ionic compounds in solution.
- Non-metal ionic elements or compounds will be collected at the anode where they lose their extra electrons.
- Metal ions less reactive than Hydrogen will be collected at the cathode where they gain electrons.